
This AOP is licensed under the BY-SA license. This license allows reusers to distribute, remix, adapt, and build upon the material in any medium or format, so long as attribution is given to the creator. The license allows for commercial use. If you remix, adapt, or build upon the material, you must license the modified material under identical terms.
AOP: 477
Title
Androgen receptor (AR) antagonism leading to hypospadias in male (mammalian) offspring
Short name
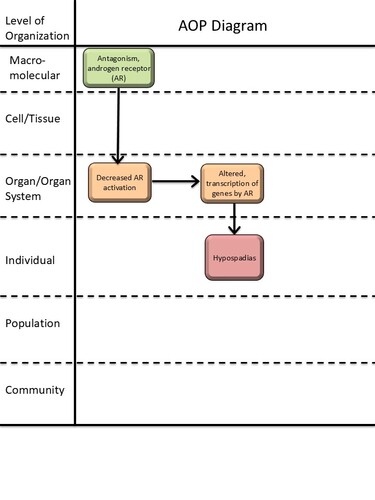
Graphical Representation
Point of Contact
Contributors
- Terje Svingen
- Emilie Elmelund
Coaches
OECD Information Table
| OECD Project # | OECD Status | Reviewer's Reports | Journal-format Article | OECD iLibrary Published Version |
|---|---|---|---|---|
| Scientific Review |
This AOP was last modified on March 20, 2026 08:27
Revision dates for related pages
| Page | Revision Date/Time |
|---|---|
| Hypospadias, increased | March 20, 2026 06:37 |
| Antagonism, Androgen receptor | March 20, 2026 11:42 |
| Decrease, androgen receptor activation | March 20, 2026 11:54 |
| Altered, Transcription of genes by the androgen receptor | March 20, 2026 12:02 |
| Antagonism, Androgen receptor leads to Decrease, AR activation | March 20, 2026 12:15 |
| Decrease, AR activation leads to Hypospadias | March 20, 2026 05:58 |
| Decrease, AR activation leads to Altered, Transcription of genes by the AR | March 20, 2026 10:30 |
| Flutamide | August 14, 2025 05:22 |
| Vinclozolin | May 14, 2020 11:28 |
| Procymidone | May 18, 2020 12:55 |
Abstract
This AOP links in utero androgen receptor (AR) antagonism with hypospadias in male offspring. Hypospadias is a common reproductive disorder with a prevalence of up to ~1/125 newborn boys, though with high inter-country variability (Leunbach et al., 2025; Paulozzi, 1999). Developmental exposure to endocrine disrupting chemicals is suspected to contribute to some cases of hypospadias (Mattiske & Pask, 2021). Hypospadias can be indicative of fetal disruptions to male reproductive development, and is associated with short anogenital distance and cryptorchidism (Skakkebaek et al., 2016). Thus, hypospadias is included as a genital abnormality in OECD test guidelines (TG) for developmental and reproductive toxicity (TG 414, 416, 421/422, and 443; (OECD, 2016b, 2016a, 2018a, 2018b, 2021)), as both a measurement of adverse reproductive effects and a direct clinical adverse outcome. In normal male reproductive development, androgen activation of the AR plays an essential role in driving differentiation of the male phenotype, including development of the penis (Amato et al., 2022). This AOP delineates the evidence that antagonism of AR through a reduction in AR activation can disrupt penis development and cause hypospadias. Substantial evidence exists that links exposure to AR antagonists to hypospadias in in vivo rodent studies. Moreover, human case studies of subjects with AR mutations and hypospadias support the link. Downstream of a reduction in AR activation, the molecular mechanisms of hypospadias development are less clear, highlighting a knowledge gap in this AOP. Thus, the AOP has potential for inclusion of additional KEs and elaboration of molecular causality links, once these are established. Given that hypospadias is both a clinical and toxicological endpoint, this AOP is considered highly relevant in a regulatory context.
AOP Development Strategy
Context
This AOP is a part of an AOP network for reduced androgen receptor activation causing hypospadias in male offspring. The other AOPs in this network are AOP-570 (‘Decreased testosterone synthesis leading to hypospadias in male (mammalian) offspring’) and AOP-571 (‘5α-reductase inhibition leading to hypospadias in male (mammalian) offspring’). The purpose of the AOP network is to organize the well-established evidence for anti-androgenic mechanisms-of-action leading to hypospadias, thus informing predictive toxicology and identifying knowledge gaps for investigation and method development.
This work received funding from the European Food and Safety Authority (EFSA) under Grant agreement no. GP/EFSA/PREV/2022/01 and from the Danish Environmental Protection Agency under the Danish Center for Endocrine Disrupters (CeHoS).
Strategy
The OECD AOP Developer’s Handbook was followed alongside pragmatic approaches (Svingen et al., 2021).
KEs and upstream KER-2130 (‘antagonism, AR leads to decrease, AR activation’) and KER-2124 (‘decrease, AR activation leads to altered, transcription of genes by AR’) were considered canonical knowledge and part of an upstream anti-androgenic network developed using mainly key review articles (Draskau et al., 2024). The non-adjacent KER-2828 linking reduced AR activation with hypospadias was developed using a systematic weight-of-evidence approach, following methodology outlined in (Holmer et al., 2024). Articles were retrieved by literature searches in PubMed and Web of Science and extensive screening using pre-defined inclusion and exclusion criteria. Evaluation of methodological reliability of in vivo animal studies was performed using the Science in Risk Assessment and Policy (SciRAP) online tool. As there are currently no in vivo methods to measure AR activation in mammals, six chemicals with known anti-androgenic mechanisms-of-action were chosen for the empirical evidence for this KER. To supplement the in vivo toxicity studies, human case studies and epidemiological studies were included in the KER. These studies were not systematically evaluated for reliability but served as supporting evidence.
Regarding the inclusion of KEs and KERs, the rationale for the upstream anti-androgenic network is detailed in (Draskau et al., 2024). The link between the upstream network, more specifically KE-286 (‘altered, transcription of genes by AR’), and AO-2082 (‘hypospadias’) likely contains a tissue-specific KE that has not been developed, as sufficient evidence is not yet available. Thus, for now, the strongest evidence linking the upstream anti-androgenic network to hypospadias is captured by KER-2828.
Summary of the AOP
Events:
Molecular Initiating Events (MIE)
Key Events (KE)
Adverse Outcomes (AO)
| Type | Event ID | Title | Short name |
|---|
| MIE | 26 | Antagonism, Androgen receptor | Antagonism, Androgen receptor |
| KE | 1614 | Decrease, androgen receptor activation | Decrease, AR activation |
| KE | 286 | Altered, Transcription of genes by the androgen receptor | Altered, Transcription of genes by the AR |
| AO | 2082 | Hypospadias, increased | Hypospadias |
Relationships Between Two Key Events (Including MIEs and AOs)
| Title | Adjacency | Evidence | Quantitative Understanding |
|---|
| Antagonism, Androgen receptor leads to Decrease, AR activation | adjacent | High | |
| Decrease, AR activation leads to Altered, Transcription of genes by the AR | adjacent | High |
| Decrease, AR activation leads to Hypospadias | non-adjacent | High |
Network View
Prototypical Stressors
Life Stage Applicability
| Life stage | Evidence |
|---|---|
| Foetal | High |
Taxonomic Applicability
Sex Applicability
| Sex | Evidence |
|---|---|
| Male | High |
Overall Assessment of the AOP
Domain of Applicability
Although the upstream part of the AOPN has a broad applicability domain, the overall AOPN is considered only applicable to male mammals during fetal life, restricted by the applicability of KER-2828 (‘decrease, AR activation leads to hypospadias’). The term hypospadias is mainly used for describing malformation of the male, and not female, external genitalia. Some studies refer to hypospadias in females, but these have not been reported to be caused by exposure to AR antagonists, and the mechanisms behind these malformations are likely different from the mechanisms in males (Greene, 1937; Stewart et al., 2018). The genital tubercle is programmed by androgens to differentiate into a penis in fetal life during the masculinization programming window, followed by the morphological differentiation (Welsh et al., 2008). In humans, hypospadias is diagnosed at birth and can also often be observed in rodents (rats and mice) at this time point, although the rodent penis does not finish developing until a few weeks after birth (Baskin & Ebbers, 2006; Sinclair et al., 2017). The disruption to androgen programming leading to hypospadias thus takes place during fetal life, but the AO itself is best detected postnatally. Specifically, the masculinization programming window (~gestational days (GD) 16-20 in rat, presumably gestational weeks (GW) 8-14 in humans) is the primary fetal window of applicability, but effects outside of this window in fetal life, after androgen production has started, cannot be excluded. Regarding taxonomic applicability, hypospadias has mainly been described in rodents and humans, and the evidence in this AOP is almost exclusively from these species. It is, however, biologically plausible that the AOP is applicable to other mammals, given the conserved role of androgens in mammalian reproductive development, and hypospadias has been observed in many domestic animal and wildlife species, albeit not coupled to AR antagonism.
Essentiality of the Key Events
|
Event |
Evidence |
Uncertainties and inconsistencies |
|
MIE-26 Antagonism, AR (high) |
Biological plausibility provides strong support for the essentiality of this event, as androgens, acting through AR, are the primary drivers of penis development. In vivo toxicity studies in rats show that in utero exposure to known AR antagonists flutamide, procymidone, and vinclozolin cause hypospadias in male offspring (listed in Table 3 in KER-2828). Ex vivo culture of genital tubercles with AR antagonist flutamide disrupted genital tubercle development, but this was rescued with addition of DHT (Petiot et al., 2005). |
In the ex vivo study, low levels of androgens were present in baseline culture media through 10% fetal calf serum supplement, and it is uncertain whether such levels are sufficient to induce in vivo-like penis differentiation. |
|
KE-1614 Decrease, AR activation (moderate) |
Biological plausibility provides strong support for the essentiality of this event, as AR activation is critical for normal penis development.
Conditional or full knockout of Ar in mice results in partly or full sex-reversal of males, including a female-like urethral opening(Willingham et al., 2006; Yucel et al., 2004; Zheng et al., 2015). Human subjects with AR mutations may also have associated hypospadias (as listed in Table 4 in KER-2828). |
|
|
KE-286 Altered, transcription of genes by AR (moderate) |
Biological plausibility provides support for the essentiality of this event. AR is a nuclear receptor and transcription factor regulating transcription of genes, and androgens, acting through AR, are essential for normal male penis development.Genomic AR signaling is necessary and possibly sufficient to drive penis differentiation (Cunha et al., 2021) Known AR-responsive genes active in normal penis development have been thoroughly reviewed (Amato et al., 2022). |
There are currently no AR-responsive genes proved to be causally involved in hypospadias, and it is known that the AR can also signal through non-genomic actions, though these alone are not sufficient for penis differentiation –and genomic AR signaling is necessary for penis development (Cunha et al., 2021; Leung & Sadar, 2017). |
|
Event |
Direct evidence |
Indirect evidence |
Contradictory evidence |
Overall essentiality assessment |
|
MIE-26 |
*** |
|
|
High |
|
KE-1614 |
** |
|
|
Moderate |
|
KE-286 |
** |
* |
|
Moderate |
Evidence Assessment
The confidence in each of the KERs comprising the AOP is judged as high, with both high biological plausibility and high confidence in the empirical evidence. The mechanistic link between KE-286 (‘altered, transcription of genes by AR’) and AO-2082 (‘hypospadias’) is not established, but given the high confidence in the KERs including the non-adjacent KER-2828 linking to the AO, the overall confidence in the AOP is judged as high.
|
KER |
Biological Plausibility |
Empirical Evidence |
Rationale |
|
KER-2130 Antagonism, AR leads to decrease, AR activation |
High |
High (canonical) |
It is well established that antagonism of the AR leads to decreased AR activity, and this has been evidenced in vitro (Draskau et al., 2024; Pedersen et al., 2022). |
|
KER-2124 Decrease, AR activation leads to altered, transcription of genes by AR |
High |
High (canonical) |
It is well established that the AR regulates gene transcription. In vivo animal studies and human genomic profiling show tissue-specific changes to gene expression upon disruption of AR (Draskau et al., 2024). |
|
KER-2828 Decrease, AR activation leads to hypospadias |
High |
High |
It is well established that AR drives penis differentiation. Numerous in vivo toxicity studies and human case studies indirectly show that decreased AR activation leads to hypospadias, with few inconsistencies. The empirical evidence moderately supports temporal concordance for the KER. It should be recognized that the upstream KE-1614 cannot currently be measured directly (in vivo). Instead, empirical evidence was therefore collected for substances known to affect upstream events. This limitation is not considered to lower the strength of the evidence in this case. |
Known Modulating Factors
|
Modulating factor (MF) |
Influence or Outcome |
KER(s) involved |
|
Genotype |
Extended CAG repeat length in AR is associated with reduced AR activity (Chamberlain et al., 1994; Tut et al., 1997). This MF could initiate the AOP at lower stressor doses. |
KER-2124, KER-2828 |
Quantitative Understanding
The quantitative understanding of this AOP is judged as low.
Considerations for Potential Applications of the AOP (optional)
References
Amato, C. M., Yao, H. H.-C., & Zhao, F. (2022). One Tool for Many Jobs: Divergent and Conserved Actions of Androgen Signaling in Male Internal Reproductive Tract and External Genitalia. Frontiers in Endocrinology, 13, 910964. https://doi.org/10.3389/fendo.2022.910964
Baskin, L., & Ebbers, M. (2006). Hypospadias: Anatomy, etiology, and technique. Journal of Pediatric Surgery, 41(3), 463–472. https://doi.org/10.1016/j.jpedsurg.2005.11.059
Chamberlain, N. L., Driver, E. D., & Miesfeld, R. L. (1994). The length and location of CAG trinucleotide repeats in the androgen receptor N-terminal domain affect transactivation function. Nucleic Acids Research, 22(15), 3181–3186. https://doi.org/10.1093/nar/22.15.3181
Cunha, G. R., Cao, M., Derpinghaus, A., Baskin, L. S., Cooke, P., & Walker, W. (2021). Cornification and classical versus nonclassical androgen receptor signaling in mouse penile/preputial development. Differentiation, 121, 1–12. https://doi.org/10.1016/j.diff.2021.08.002
Draskau, M., Rosenmai, A., Bouftas, N., Johansson, H., Panagiotou, E., Holmer, M., Elmelund, E., Zilliacus, J., Beronius, A., Damdimopoulou, P., van Duursen, M., & Svingen, T. (2024). Aop Report: An Upstream Network for Reduced Androgen Signalling Leading to Altered Gene Expression of Ar Responsive Genes in Target Tissues. Environ Toxicol Chem, (In Press).
Greene, R. R. (1937). Production of Experimental Hypospadias in the Female Rat. Proceedings of the Society for Experimental Biology and Medicine, 36(4), 503–506. https://doi.org/10.3181/00379727-36-9287P
Holmer, M. L., Zilliacus, J., Draskau, M. K., Hlisníková, H., Beronius, A., & Svingen, T. (2024). Methodology for developing data-rich Key Event Relationships for Adverse Outcome Pathways exemplified by linking decreased androgen receptor activity with decreased anogenital distance. Reproductive Toxicology, 128, 108662. https://doi.org/10.1016/j.reprotox.2024.108662
Leunbach, T. L., Berglund, A., Ernst, A., Hvistendahl, G. M., Rawashdeh, Y. F., & Gravholt, C. H. (2025). Prevalence, Incidence, and Age at Diagnosis of Boys With Hypospadias: A Nationwide Population-Based Epidemiological Study. Journal of Urology, 213(3), 350–360. https://doi.org/10.1097/JU.0000000000004319
Leung, J. K., & Sadar, M. D. (2017). Non-Genomic Actions of the Androgen Receptor in Prostate Cancer. Frontiers in Endocrinology, 8. https://doi.org/10.3389/fendo.2017.00002
Mattiske, D. M., & Pask, A. J. (2021). Endocrine disrupting chemicals in the pathogenesis of hypospadias; developmental and toxicological perspectives. Current Research in Toxicology, 2, 179–191. https://doi.org/10.1016/j.crtox.2021.03.004
OECD. (2001). Test No. 416: Two-Generation Reproduction Toxicity [OECD Guidelines for the Testing of Chemicals, Section 4]. OECD Publishing. https://doi.org/10.1787/9789264070868-en
OECD. (2016a). Test No. 421: Reproduction/Developmental Toxicity Screening Test. OECD. https://doi.org/10.1787/9789264264380-en
OECD. (2016b). Test No. 422: Combined Repeated Dose Toxicity Study with the Reproduction/Developmental Toxicity Screening Test. OECD. https://doi.org/10.1787/9789264264403-en
OECD. (2018a). Test No. 414: Prenatal Developmental Toxicity Study. OECD. https://doi.org/10.1787/9789264070820-en
OECD. (2018b). Test No. 443: Extended One-Generation Reproductive Toxicity Study. OECD. https://doi.org/10.1787/9789264185371-en
Paulozzi, L. J. (1999). International trends in rates of hypospadias and cryptorchidism.
Pedersen, E. B., Christiansen, S., & Svingen, T. (2022). AOP key event relationship report: Linking androgen receptor antagonism with nipple retention. Current Research in Toxicology, 3, 100085. https://doi.org/10.1016/j.crtox.2022.100085
Petiot, A., Perriton, C., Dickson, C., & Cohn, M. (2005). Development of the mammalian urethra is controlled by Fgfr2-IIIb. Development (Cambridge, England), 132(10), 2441–2450. (rayyan-427635023). https://doi.org/10.1242/dev.01778
Sinclair, A., Cao, M., Pask, A., Baskin, L., & Cunha, G. (2017). Flutamide-induced hypospadias in rats: A critical assessment. Differentiation; Research in Biological Diversity, 94, 37–57. (rayyan-427634666). https://doi.org/10.1016/j.diff.2016.12.001
Skakkebaek, N. E., Rajpert-De Meyts, E., Louis, G. M. B., Toppari, J., Andersson, A.-M., Eisenberg, M. L., Jensen, T. K., Jorgensen, N., Swan, S. H., Sapra, K. J., Ziebe, S., Priskorn, L., & Juul, A. (2016). Male Reproductive Disorders And Fertility Trends: Influences Of Environement And Genetic susceptibility. PHYSIOLOGICAL REVIEWS, 96(1), 55–97. (WOS:000370383200003). https://doi.org/10.1152/physrev.00017.2015
Stewart, M. K., Mattiske, D. M., & Pask, A. J. (2018). In utero exposure to both high- and low-dose diethylstilbestrol disrupts mouse genital tubercle development†. Biology of Reproduction, 99(6), 1184–1193. https://doi.org/10.1093/biolre/ioy142
Svingen, T., Villeneuve, D. L., Knapen, D., Panagiotou, E. M., Draskau, M. K., Damdimopoulou, P., & O’Brien, J. M. (2021). A Pragmatic Approach to Adverse Outcome Pathway Development and Evaluation. Toxicological Sciences, 184(2), 183–190. https://doi.org/10.1093/toxsci/kfab113
Tut, T. G., Ghadessy, F. J., Trifiro, M. A., Pinsky, L., & Yong, E. L. (1997). Long Polyglutamine Tracts in the Androgen Receptor Are Associated with Reduced Trans -Activation, Impaired Sperm Production, and Male Infertility 1. The Journal of Clinical Endocrinology & Metabolism, 82(11), 3777–3782. https://doi.org/10.1210/jcem.82.11.4385
Welsh, M., Saunders, P. T. K., Fisken, M., Scott, H. M., Hutchison, G. R., Smith, L. B., & Sharpe, R. M. (2008). Identification in rats of a programming window for reproductive tract masculinization, disruption of which leads to hypospadias and cryptorchidism. Journal of Clinical Investigation, 118(4), 1479–1490. https://doi.org/10.1172/JCI34241
Willingham, E., Agras, K., Souza, A. J. de, Konijeti, R., Yucel, S., Rickie, W., Cunha, G., & Baskin, L. (2006). Steroid receptors and mammalian penile development: An unexpected role for progesterone receptor? The Journal of Urology, 176(2), 728–733. (rayyan-427634662). https://doi.org/10.1016/j.juro.2006.03.078
Yucel, S., Liu, W., Cordero, D., Donjacour, A., Cunha, G., & Baskin, L. (2004). Anatomical studies of the fibroblast growth factor-10 mutant, Sonic Hedge Hog mutant and androgen receptor mutant mouse genital tubercle. Advances in Experimental Medicine and Biology, 545, 123–148. (rayyan-427634903). https://doi.org/10.1007/978-1-4419-8995-6_8
Zheng, Z., Armfield, B., & Cohn, M. (2015). Timing of androgen receptor disruption and estrogen exposure underlies a spectrum of congenital penile anomalies. Proceedings of the National Academy of Sciences of the United States of America, 112(52), E7194-203. (rayyan-427635801). https://doi.org/10.1073/pnas.1515981112