
This AOP is licensed under the BY-SA license. This license allows reusers to distribute, remix, adapt, and build upon the material in any medium or format, so long as attribution is given to the creator. The license allows for commercial use. If you remix, adapt, or build upon the material, you must license the modified material under identical terms.
AOP: 609
Title
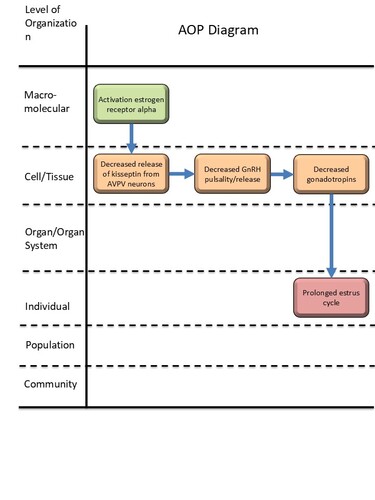
Activation, estrogen receptor alpha leads to prolonged estrus cycle via decreased kisspeptin release
Short name
Graphical Representation
Point of Contact
Contributors
- John Frisch
Coaches
OECD Information Table
| OECD Project # | OECD Status | Reviewer's Reports | Journal-format Article | OECD iLibrary Published Version |
|---|---|---|---|---|
This AOP was last modified on March 18, 2026 16:03
Revision dates for related pages
| Page | Revision Date/Time |
|---|---|
| Activation, estrogen receptor alpha | January 28, 2026 14:32 |
| Decreased, release of kisspeptin from anteroventral periventricular nucleus (AVPV) neurons | April 06, 2026 14:39 |
| Decreased, GnRH pulsatility/release | January 28, 2026 14:39 |
| Decreased, Gonadotropins | April 06, 2026 14:41 |
| prolonged, estrus cycle | October 03, 2025 11:27 |
| Activation, ERα leads to Decreased, release of kisspeptin from AVPV neurons | March 18, 2026 16:07 |
| Decreased, release of kisspeptin from AVPV neurons leads to Decreased, GnRH pulsatility/release | January 28, 2026 15:31 |
| Decreased, GnRH pulsatility/release leads to prolonged, estrus cycle | January 28, 2026 15:02 |
| Decreased, Gonadotropins leads to prolonged, estrus cycle | January 28, 2026 15:05 |
Abstract
Estrogen receptor alpha (ERa) is a nuclear transcription factor involved in regulation of many physiological processes in mammals. Binding by estrogen induces the transcription of target genes. Here we focus on the role of ERa in the hypothalamus- pituitary-gonadal (HPG) axis involved in reproductive development and the estrus cycle through activation of kisspeptin.
Kisspeptin is a key signalling neuropeptide hormone in mammals. Positive feedback for kisspeptin hormone production is due to increased levels of estrogen binding to Estrogen Receptor Alpha (ERa) receptors in neurons from the anteroventral periventricular nucleus (AVPV) region of the hypothalamus, while negative feedback for kisspeptin hormone production is due to ERa receptor activation of the neurons from the arcuate nucleus (ARC) region of the hypothalamus (Uenoyama et al. 2021). Kisspeptin signalling is important for prompting hormone production for coordinating the estrus cycle.
Gonadotropin-releasing hormone (GnRH) is produced by the hypothalamus. Increases in GnRH stimulates increased production of gonadotropins. Luteinizing hormone (LH) and Follicle-stimulating hormone (FSH) are gonadotropins of particular interest because of their roles in regulating gonadal steroid biosynthesis, development of reproductive organs, and the estrus cycle. Decreased Gonadotropin-releasing, luteinizing hormone, and follicle-stimulating hormone levels and/or release frequency leads to issues in reproductive development (Casarini and Simoni 2021; Howard 2021) and abnormal estrus cycles (Uenoyama et al. 2021).
The estrus cycle is a coordinated series of changes that results in fertility in mammals. Changes to the uterus and vagina are coordinated through hormone signaling, including Progesterone, Estradiol, Luteinizing Hormone, and Follicle-Stimulating Hormone in order to progress through metestrus, diestrus, proestrus, and estrous phases (Miller and Takahashi 2014; Swift et al. 2014). Prolonged estrus occurs when the normal estrus cycle progression has been disrupted, generally through increased diestrus.
This AOP links ERa activation to prolonged estrus as one of the adverse outcomes observed in Endocrine Disruptor Screening Program (EDSP) protocol (US EPA 1998, 2011; OECD 2025).
AOP Development Strategy
Context
This AOP was part of an Environmental Protection Agency effort to develop AOPs that establish scientifically supported causal linkages between alternative endpoints measured using new approach methodologies (NAMs) and guideline apical endpoints measured in Tier 1 and Tier 2 test guidelines (U.S. EPA, 2024) employed by the Endocrine Disruptor Screening Program (EDSP). A series of key events that represent significant, measurable, milestones connecting molecular initiation to apical endpoints indicative of adversity were identified based on scientific review articles and empirical studies. Additionally, scientific evidence supporting the causal relationships between each pair of key events was assembled and evaluated. The present effort focused primarily on empirical studies with laboratory rodents and other mammals.
Strategy
The scope of the aforementioned EPA project was to develop AOP(s) relevant to apical endpoints observed in the test guidelines, based on mechanisms consistent with empirical studies. The literature used to support this AOP and its constituent pages began with the test guidelines and followed to primary, secondary, and/or tertiary works concerning the relevant underlying biology. KE and KER page creation and re-use was determined using Handbook principles where page re-use was preferred.

Summary of the AOP
Events:
Molecular Initiating Events (MIE)
Key Events (KE)
Adverse Outcomes (AO)
| Type | Event ID | Title | Short name |
|---|
| MIE | 1065 | Activation, estrogen receptor alpha | Activation, ERα |
| KE | 968 | Decreased, release of kisspeptin from anteroventral periventricular nucleus (AVPV) neurons | Decreased, release of kisspeptin from AVPV neurons |
| KE | 530 | Decreased, GnRH pulsatility/release | Decreased, GnRH pulsatility/release |
| KE | 1986 | Decreased, Gonadotropins | Decreased, Gonadotropins |
| AO | 1075 | prolonged, estrus cycle | prolonged, estrus cycle |
Relationships Between Two Key Events (Including MIEs and AOs)
| Title | Adjacency | Evidence | Quantitative Understanding |
|---|
Network View
Prototypical Stressors
Life Stage Applicability
| Life stage | Evidence |
|---|---|
| Adult, reproductively mature | Moderate |
| Juvenile | Moderate |
Taxonomic Applicability
| Term | Scientific Term | Evidence | Link |
|---|---|---|---|
| mammals | mammals | Moderate | NCBI |
Sex Applicability
| Sex | Evidence |
|---|---|
| Female | High |
Overall Assessment of the AOP
|
1. Support for Biological Plausibility of Key Event Relationships: Is there a mechanistic relationship between KEup and KEdown consistent with established biological knowledge? |
|
|
Key Event Relationship (KER) |
Level of Support Strong = Extensive understanding of the KER based on extensive previous documentation and broad acceptance. Moderate = Support of the relationship based on empirical studies, with some inference of receptor activation in laboratory mammals from in vitro studies. |
|
Relationship 3646: Activation estrogen receptor alpha leads to decreased AVPV kisspeptin release |
Moderate support. The relationship between activation of estrogen receptor alpha and decreased AVPV kisspeptin release is broadly accepted and supported among humans and laboratory mammal data. Activation of estrogen receptor alpha is often studied in vitro, with activation of estrogen receptor alpha inferred in laboratory mammal studies when downstream effects are consistent with in vitro observations. Activation of estrogen receptor alpha can lead to either increase or decrease of AVPV kisspeptin release depending on the stressor. Broadly, neonatal developmental stressor exposure can disrupt the Hypothalamic-Pituitary-Gonadal axis, decreasing AVPV kisspeptin release and subsequently decreasing hormone levels (Bateman and Patisaul 2008; Homma et al. 2009; Navarro et al. 2009; Patisaul et al. 2009; Ichimura et al. 2015a; Ichimura et al. 2015b), dampening response to hormones. Alternatively, stressor exposure can lead to increased AVPV kisspeptin release and subsequent increased hormone levels (Adachi et al. 2007; Clarkson et al. 2008; Tomikawa et al. 2012; Wang et al. 2014), accelerating the response to hormones in the expected direction from estrogen receptor alpha activation to increased AVPV kisspeptin release. |
|
Relationship 3647: Decreased AVPV kisspeptin release leads to decreased GnRH pulsatility/release. |
Strong support. The relationship between decreased AVPV kisspeptin release and decreased GnRH release is broadly accepted and supported among humans and laboratory mammal data. |
|
Relationship 3648: Decreased GnRH pulsatility/release leads to decreased gonadotropins |
Strong support. The relationship between decreased GnRH release and decreased gonadotropins is broadly accepted and supported among humans and laboratory mammal data. |
|
Relationship 3649: Decreased gonadotropins leads to prolonged estrus cycle |
Strong support. The relationship between decreased gonadotropins and prolonged estrus is broadly accepted and supported among humans and laboratory mammal data. |
|
Overall |
Moderate to Strong support. Extensive understanding of the relationships between events from empirical studies from humans and laboratory mammals, with some inference of estrogen receptor alpha activation from in vitro studies when performing laboratory mammal studies. |
Domain of Applicability
Life Stage: Applies to adult, reproductively mature and juveniles.
Sex: Applies to females.
Taxonomic: Primarily studied in laboratory rodents (have an estrus cycle) and humans (have a menstrual cycle which differs from estrus cycle by shedding of uterine lining). Plausible for most mammals due to conserved hormone pathways regulating hypothalamus-pituitary-gonadal axis processes. For vertebrates, kisspeptin and kisspeptin receptors are absent from birds; the relationship between estrogen and kisspeptin is also unclear for fish and may play a compensatory rather than one required for normal endocrine cycling (Sivalingam et al 2022). GnRH and gonadotropins are widespread among amphibians, reptiles, fish, birds, and mammals (Duan and Allard 2020; Hollander-Cohen et al. 2021).
Essentiality of the Key Events
|
2. Essentiality of Key Events: Are downstream KEs and/or the AO prevented if an upstream KE is blocked? |
|
|
Key Event (KE) |
Level of Support Strong = Direct evidence from specifically designed experimental studies illustrating essentiality and direct relationship between key events. |
|
MIE 1065 Activation estrogen receptor alpha |
Strong support. Activation of estrogen receptor alpha leads to decreased AVPV kisspeptin release. Evidence is available from toxicant studies and gene-knock out studies with in vitro human cell lines and intact laboratory mammals. Best evidence for essentiality for activation of estrogen receptor alpha is baseline levels of kisspeptin in the absence of stressor. Activation of estrogen receptor alpha can lead to either increase or decrease of AVPV kisspeptin release depending on the stressor. |
|
KE 968 Decreased AVPV kisspeptin release |
Strong support. Decreased AVPV kisspeptin release leads to decreased GnRH pulsality/release. Evidence is available from toxicant studies, gene-knock out studies, and ovariectomized animal studies. Best evidence for essentiality for decreased AVPV release is in stressor studies with observed decreased GnRH hormone levels, and restored GnRH levels from supplemental addition of kisspeptin. |
|
KE 530 Decreased GnRH pulsatility/release |
Strong support. Decreased GnRH pulsatility/release leads to decreased gonadotropins. Evidence is available from toxicant studies and ovariectomized animal studies. Best evidence for essentiality for decreased GnRH pulsatility/release is in hormone replacement studies in which normal gonadotropin levels are restored from GnRH addition to animals with low GnRH levels from a stressor. |
|
KE 1986 Decreased Gonadotropins |
Strong support. Decreased gonadotropins leads to prolonged estrus. Evidence is available from toxicant studies and ovariectomized animal studies. Best evidence for essentiality for decreased gonadotropins is from hormone replacement studies in which normal estrus cycles are restored after administration of exogenous gonadotropins to animals that were exhibiting prolonged estrus and low gonadotropin levels after exposure to a stressor. |
|
AO 1075 Prolonged estrus cycle |
This is the final event of the AOP. |
|
Overall |
Strong support. Direct evidence from empirical studies from laboratory mammals and human cell lines for all key events. |
Evidence Assessment
|
3. Empirical Support for Key Event Relationship: Does empirical evidence support that a change in KEup leads to an appropriate change in KEdown? |
|
|
Key Event Relationship (KER) |
Level of Support Strong = Experimental evidence from exposure to toxicant shows consistent change in both events across taxa and study conditions. |
|
Relationship 3646: Activation estrogen receptor alpha leads to decreased AVPV kisspeptin release |
Strong support. Activation of estrogen receptor alpha leads to decreased AVPV kisspeptin release. Evidence is available from toxicant studies and gene-knock out studies. Activation of estrogen receptor alpha occurred earlier in the time-course of exposure decreased AVPV kisspeptin release, and the concentrations that Activated estrogen receptor alpha were equal to or lower than the concentrations that decreased AVPV kisspeptin release. Therefore, the data support a causal relationship. In some in vivo laboratory mammal studies, activation of estrogen receptor alpha is inferred by kisspeptin response from a stressor known to be an ERa agonist from in vitro studies. |
|
Relationship 3647: Decreased AVPV kisspeptin release leads to decreased GnRH pulsatility/release. |
Strong support. Decreased AVPV kisspeptin release leads to decreased GnRH pulsatility/release. Evidence is available from toxicant studies, gene-knock out studies, and ovariectomized animal studies. Decreased AVPV kisspeptin release occurred earlier in the time-course of exposure than loss of decreased GnRH pulsatility/release, and the concentrations that decreased AVPV kisspeptin release were equal to or lower than the concentrations that decreased GnRH pulsatility/release. Therefore, the data support a causal relationship. |
|
Relationship 3648: Decreased GnRH pulsatility/release leads to decreased gonadotropins |
Strong support. Decreased GnRH pulsatility/release leads to decreased gonadotropins. Evidence is available from toxicant studies and ovariectomized animal studies. Decreased GnRH pulsatility/release occurred earlier in the time-course of exposure than decreased gonadotropins, and the concentrations that Decreased GnRH pulsatility/release were equal to or lower than the concentrations that decreased gonadotropins. Therefore, the data support a causal relationship. |
|
Relationship 3649: Decreased gonadotropins leads to prolonged estrus cycle |
Strong support. Decreased gonadotropins leads to prolonged estrus. Evidence is available from toxicant studies and ovariectomized animal studies. Decreased gonadotropins occurred earlier in the time-course of exposure than prolonged estrus, and the concentrations that Decreased gonadotropins were equal to or lower than the concentrations that prolonged estrus. Therefore, the data support a causal relationship. |
|
Overall |
Strong support. Evidence from empirical studies shows consistent change relationships in upstream and downstream events, with upstream events occurring earlier in the time-course of exposure and at equal or lower concentrations than downstream events, supporting causal relationships. |
Known Modulating Factors
| Modulating Factor (MF) | Influence or Outcome | KER(s) involved |
|---|---|---|
Quantitative Understanding
Considerations for Potential Applications of the AOP (optional)
References
Adachi S, Yamada S, Takatsu Y, Matsui H, Kinoshita M, Takase K, Sugiura H, Ohtaki T, Matsumoto H, Uenoyama Y, Tsukamura H, Inoue K, Maeda K. 2007. Involvement of anteroventral periventricular metastin/kisspeptin neurons in estrogen positive feedback action on luteinizing hormone release in female rats. Journal of Reproduction and Development 53(2): 367-378.
Bateman HL, Patisaul HB. 2008. Disrupted female reproductive physiology following neonatal exposure to phytoestrogens or estrogen specific ligands is associated with decreased GnRH activation and kisspeptin fiber density in the hypothalamus. Neurotoxicology 29(6): 988-997.
Casarini L, Simoni M. 2021. Recent advances in understanding gonadotropin signaling. Faculty Reviews 10: 41.
Clarkson J, d’Anglemont de Tassigny X, Moreno AS, Colledge WH, Herbison AE. 2008. Kisspeptin–GPR54 signaling is essential for preovulatory gonadotropin-releasing hormone neuron activation and the luteinizing hormone surge. Journal of Neuroscience 28(35): 8691–8697.
Duan C, Allard J. 2020. Gonadotropin-releasing hormone neuron development in vertebrates. General and Comparative Endocrinology. 292: 113465.
Hollander-Cohen L, Golan M, Levavi-Sivan B. 2021. Differential Regulation of Gonadotropins as Revealed by Transcriptomes of Distinct LH and FSH Cells of Fish Pituitary. International Journal of Molecular Sciences 22(12): 6478.
Homma T, Sakakibara M, Yamada S, Kinoshita M, Iwata K, Tomikawa J, Kanazawa T, Matsui H, Takatsu Y, Ohtaki T, Matsumoto H, Uenoyama Y, Maeda K, Tsukamura H. 2009. Significance of neonatal testicular sex steroids to defeminize anteroventral periventricular kisspeptin neurons and the GnRH/LH surge system in male rats. Biology of Reproduction 81(6): 1216-25.
Howard SR. 2021. Interpretation of reproductive hormones before, during and after the pubertal transition—identifying health and disordered puberty. Clinical Endocrinolology 95: 702-715.
Ichimura R, Takahashi M, Morikawa T, Inoue K, Maeda J, Usuda K, Yokosuka M, Watanabe G, Yoshida M. 2015a. Prior attenuation of KiSS1/GPR54 signaling in the anteroventral periventricular nucleus is a trigger for the delayed effect induced by neonatal exposure to 17alpha-ethynylestradiol in female rats. Reproductive Toxicology 51: 145-156.
Ichimura R, Takahashi M, Morikawa T, Inoue K, Kuwata K, Usuda K, Yokosuka M, Watanabe G, Yoshida M. 2015b. The Critical Hormone-Sensitive Window for the Development of Delayed Effects Extends to 10 Days after Birth in Female Rats Postnatally Exposed to 17alpha-Ethynylestradiol. Biology of Reproduction 93(2): 32.
Miller BH, Takahashi JS. 2014. Central circadian control of female reproductive function. Frontiers in Endocrinology 4(1): 195.
Navarro VM, Sánchez-Garrido MA, Castellano JM, Roa J, García-Galiano D, Pineda R, Aguilar E, Pinilla L, Tena-Sempere M. 2009. Persistent impairment of hypothalamic KiSS-1 system after exposures to estrogenic compounds at critical periods of brain sex differentiation. Endocrinology. 150(5): 2359-2367.
Organisation for Economic Co-operation and Development. 2025. Test No. 443: Extended One-Generation Reproductive Toxicity Study, OECD Guidelines for the Testing of Chemicals, Section 4, OECD Publishing, Paris. https:// https://www.oecd.org/en/publications/test-no-443-extended-one-generation-reproductive-toxicity-study_9789264185371-en.html (retrieved 11 Dec 2025)
Patisaul HB, Todd KL, Mickens JA, Adewale HB. 2009. Impact of neonatal exposure to the ERalpha agonist PPT, bisphenol-A or phytoestrogens on hypothalamic kisspeptin fiber density in male and female rats. Neurotoxicology. 30(3): 350-357.
Sivalingam M, Ogawa S, Trudeau VL, Parhar IS. 2022. Conserved functions of hypothalamic kisspeptin in vertebrates. General and Comparative Endocrinology 317: 113973. Swift KM, Gary NC, Urbanczyk PJ. 2024. On the basis of sex and sleep: the influence of the estrous cycle and sex on sleep-wake behavior. Frontiers in Neuroscience 18:1426189.
Tomikawa J, Uenoyama Y, Ozawa M, Fukanuma T, Takase K, Goto T, Abe H, Ieda N, Minabe S, Deura C, Inoue N, Sanbo M, Tomita K, Hirabayashi M, Tanaka S, Imamura T, Okamura H, Maeda K, Tsukamura H. 2012. Epigenetic regulation of Kiss1 gene expression mediating estrogen-positive feedback action in the mouse brain. Proceedings of the National Academy of Science 109(20): E1294-E1301.
Uenoyama, Y., Inoue, N., Nakamura, S., and Tsukamura, H. Kisspeptin Neurons and Estrogen–Estrogen Receptor α Signaling: Unraveling the Mystery of Steroid Feedback System Regulating Mammalian Reproduction. 2021. International Journal of Molecular Sciences 22(17): 9229.
U.S. Environmental Protection Agency. 1998. Health Effects Test Guidelines OPPTS 870.3800 Reproduction and Fertility Effects. https://ntp.niehs.nih.gov/sites/default/files/iccvam/suppdocs/feddocs/epa/epa_870_3800.pdf (retrieved 24 December 2025)
U.S. Environmental Protection Agency. 2004. EDSP Test Guidelines and Guidance Document. https://www.epa.gov/test-guidelines-pesticides-and-toxic-substances/edsp-test-guidelines-and-guidance-document (retrieved 25 July 2025).
U.S. Environmental Protection Agency. 2011. Pubertal Development and Thyroid Function in Intact Juvenile/Peripubertal Female Rats Assay OCSPP Guideline 890.1450 https://www.epa.gov/sites/default/files/2015-07/documents/final_890.1450_female_pubertal_assay_sep_8.24.11.pdf (retrieved 24 December 2025)
Wang X, Chang F, Bai Y, Chen F, Zhang J, Chen L. 2014. Bisphenol A enhances kisspeptin neurons in anteroventral periventricular nucleus of female mice. Journal of Endocrinology 28(35): 201-213.