
This AOP is licensed under the BY-SA license. This license allows reusers to distribute, remix, adapt, and build upon the material in any medium or format, so long as attribution is given to the creator. The license allows for commercial use. If you remix, adapt, or build upon the material, you must license the modified material under identical terms.
AOP: 624
Title
Increased 11β-Hydroxysteroid dehydrogenase type 1 activity leading to MASLD progression via insulin resistance-associated mitochondrial dysfunction
Short name
Graphical Representation
Point of Contact
Contributors
- You Song
Coaches
- Shihori Tanabe
OECD Information Table
| OECD Project # | OECD Status | Reviewer's Reports | Journal-format Article | OECD iLibrary Published Version |
|---|---|---|---|---|
| Under Development |
This AOP was last modified on February 24, 2026 08:35
Revision dates for related pages
| Page | Revision Date/Time |
|---|---|
| Increase, 11β-Hydroxysteroid dehydrogenase type 1 activity | February 18, 2026 08:38 |
| Increase, Hepatic intracellular active glucocorticoids | February 26, 2026 06:48 |
| Increase, Glucocorticoid receptor activation | February 12, 2026 07:24 |
| Increase, Insulin resistance | February 11, 2026 05:50 |
| Increase, Adipose lipolysis | February 10, 2026 05:00 |
| Increased, Fatty acid influx | February 24, 2026 08:56 |
| Increase, Liver steatosis | February 11, 2026 05:41 |
| Increase, Hepatocellular lipotoxicity | February 10, 2026 04:40 |
| Increase, Mitochondrial dysfunction | February 11, 2026 07:06 |
| Increase, Cell injury/death | May 27, 2024 07:23 |
| Increase, Kupffer cell activation | February 11, 2026 05:16 |
| Increase, Steatohepatitis | February 24, 2026 09:13 |
| Increase, Transforming growth factor-beta signaling | February 11, 2026 05:39 |
| Increase, Hepatic stellate cell activation | February 11, 2026 07:04 |
| Increase, Collagen accumulation | February 11, 2026 06:58 |
| Increase, Liver fibrosis | February 11, 2026 05:35 |
| Increase, Regenerative nodule formation | February 10, 2026 06:47 |
| Increase, Cirrhosis | February 11, 2026 07:34 |
| Increase, 11β-HSD1 activity leads to Increase, Hepatic intracellular active GC | February 24, 2026 08:32 |
| Increase, Hepatic intracellular active GC leads to Increase, GR activation | February 24, 2026 08:32 |
| Increase, GR activation leads to Increase, Insulin resistance | February 12, 2026 07:38 |
| Increase, Insulin resistance leads to Increase, Adipose lipolysis | February 10, 2026 08:59 |
| Increase, Adipose lipolysis leads to Increased, FA Influx | February 24, 2026 08:33 |
| Increased, FA Influx leads to Increase, Liver steatosis | February 24, 2026 08:33 |
| Increase, Liver steatosis leads to Increase, Hepatocellular lipotoxicity | February 10, 2026 08:59 |
| Increase, Mitochondrial dysfunction leads to Cell injury/death | November 29, 2016 20:08 |
| Cell injury/death leads to Increase, Kupffer cell activation | November 29, 2016 19:54 |
| Increase, Kupffer cell activation leads to Increase, Steatohepatitis | February 10, 2026 09:00 |
| Increase, Steatohepatitis leads to Activation of TGF-β signaling | February 10, 2026 09:00 |
| Activation of TGF-β signaling leads to Increase, HSC activation | February 10, 2026 09:01 |
| Increase, HSC activation leads to Increase, Collagen accumulation | December 05, 2018 08:51 |
| Increase, Collagen accumulation leads to Increase, Liver fibrosis | December 05, 2018 08:52 |
| Increase, Liver fibrosis leads to Increase, Regenerative nodule formation | February 10, 2026 09:02 |
| Increase, Regenerative nodule formation leads to Increase, Cirrhosis | February 10, 2026 09:02 |
Abstract
This adverse outcome pathway (AOP) describes a mechanistically coherent sequence linking altered glucocorticoid receptor (GR) signaling to the progression of metabolic dysfunction–associated steatotic liver disease (MASLD), including steatosis, steatohepatitis (MASH), fibrosis, and cirrhosis. The pathway integrates endocrine disruption–induced perturbations in GR signaling with systemic insulin resistance, dysregulated adipose lipolysis, increased hepatic free fatty acid (FFA) influx, hepatocellular lipotoxicity, mitochondrial dysfunction, oxidative stress, and inflammatory and fibrogenic responses within the liver. Downstream activation of Kupffer cells, hepatic stellate cells, and TGF-β signaling drives extracellular matrix deposition and fibrotic remodeling, culminating in advanced liver disease outcomes. This AOP is particularly relevant for the identification and prioritization of endocrine-disrupting chemicals (EDCs) that interfere with GR signaling and contribute to MASLD progression, supporting its application in screening, hazard identification, and the development of integrated testing strategies.
AOP Development Strategy
Context
MASLD is a highly prevalent chronic liver disease driven by metabolic dysfunction, insulin resistance, and lipotoxic stress, with increasing evidence that endocrine signaling perturbations play a critical upstream role. Glucocorticoid receptor (GR) signaling is central to the regulation of glucose and lipid metabolism, adipose tissue lipolysis, mitochondrial function, and inflammatory responses. Dysregulation of GR signaling—whether through chronic activation, impaired feedback control, or chemical interference—has been linked to systemic insulin resistance and altered lipid fluxes that place a metabolic burden on the liver.
This AOP was developed to capture the biological plausibility and mechanistic continuity between altered GR signaling and MASLD progression, with a specific emphasis on insulin resistance–associated mitochondrial dysfunction as a unifying driver of disease advancement. The pathway reflects clinically relevant disease stages and aligns with regulatory interest in identifying upstream endocrine-mediated mechanisms contributing to chronic metabolic liver disease.
Strategy
The development of this AOP was informed by expert-driven scoping combined with targeted literature evaluation across endocrinology, metabolic disease, toxicology, and hepatology. Initial pathway framing was based on well-established physiological roles of GR signaling in metabolic homeostasis and on clinical and experimental evidence linking glucocorticoid dysregulation to insulin resistance and fatty liver disease.
Focused literature searches were conducted to identify evidence supporting individual key events (KEs) and key event relationships (KERs), including:
-
Altered GR signaling and insulin resistance
-
Adipose tissue lipolysis and hepatic FFA influx
-
Hepatocellular lipotoxicity and mitochondrial dysfunction
-
Oxidative stress, cell injury, and inflammatory activation
-
Fibrogenic signaling pathways and disease stage progression
Priority was given to peer-reviewed human, animal, and mechanistic in vitro studies demonstrating causal or concordant relationships between upstream and downstream events. Evidence was screened for biological plausibility, consistency across models, and relevance to regulatory toxicology and EDC assessment.
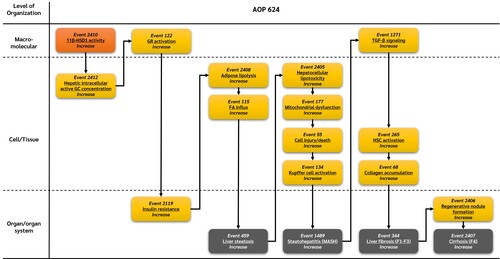
Summary of the AOP
Events:
Molecular Initiating Events (MIE)
Key Events (KE)
Adverse Outcomes (AO)
| Type | Event ID | Title | Short name |
|---|
| MIE | 2410 | Increase, 11β-Hydroxysteroid dehydrogenase type 1 activity | Increase, 11β-HSD1 activity |
| KE | 2412 | Increase, Hepatic intracellular active glucocorticoids | Increase, Hepatic intracellular active GC |
| KE | 122 | Increase, Glucocorticoid receptor activation | Increase, GR activation |
| KE | 2119 | Increase, Insulin resistance | Increase, Insulin resistance |
| KE | 2408 | Increase, Adipose lipolysis | Increase, Adipose lipolysis |
| KE | 465 | Increased, Fatty acid influx | Increased, FA Influx |
| KE | 2405 | Increase, Hepatocellular lipotoxicity | Increase, Hepatocellular lipotoxicity |
| KE | 177 | Increase, Mitochondrial dysfunction | Increase, Mitochondrial dysfunction |
| KE | 55 | Increase, Cell injury/death | Cell injury/death |
| KE | 134 | Increase, Kupffer cell activation | Increase, Kupffer cell activation |
| KE | 1271 | Increase, Transforming growth factor-beta signaling | Activation of TGF-β signaling |
| KE | 265 | Increase, Hepatic stellate cell activation | Increase, HSC activation |
| KE | 68 | Increase, Collagen accumulation | Increase, Collagen accumulation |
| KE | 2406 | Increase, Regenerative nodule formation | Increase, Regenerative nodule formation |
| AO | 459 | Increase, Liver steatosis | Increase, Liver steatosis |
| AO | 1489 | Increase, Steatohepatitis | Increase, Steatohepatitis |
| AO | 344 | Increase, Liver fibrosis | Increase, Liver fibrosis |
| AO | 2407 | Increase, Cirrhosis | Increase, Cirrhosis |
Relationships Between Two Key Events (Including MIEs and AOs)
| Title | Adjacency | Evidence | Quantitative Understanding |
|---|
Network View
Prototypical Stressors
Life Stage Applicability
Taxonomic Applicability
Sex Applicability
Overall Assessment of the AOP
This AOP is biologically plausible and supported by a substantial body of empirical evidence linking endocrine disruption, insulin resistance, mitochondrial dysfunction, and MASLD progression. The sequence of events reflects conserved metabolic and inflammatory processes observed across species, with strong concordance between experimental models and human disease pathology.
The AOP is well-suited for regulatory applications focused on hazard identification and prioritization, particularly for chemicals that modulate GR signaling but may not induce overt liver toxicity in short-term assays. While quantitative understanding varies across individual KERs, the overall weight of evidence supports the use of this AOP in integrated testing and assessment frameworks rather than as a stand-alone risk assessment tool.
Domain of Applicability
-
Taxa: Primarily mammals (rodents and humans)
-
Life stage: Adolescents and adults (relevance highest in metabolically mature organisms)
-
Sex: Both sexes; sex-specific sensitivity may occur due to hormonal and metabolic differences
-
Biological context: Metabolic dysfunction, obesity, insulin resistance, and chronic endocrine perturbation
The AOP is most applicable under conditions of sustained metabolic stress and is not intended to represent acute liver injury mechanisms.
Essentiality of the Key Events
Essentiality of the KEs is supported by both direct and indirect evidence demonstrating that modulation of upstream events alters the occurrence or severity of downstream outcomes:
-
Altered GR signaling: Experimental and clinical modulation of GR activity directly influences insulin sensitivity, lipid metabolism, and hepatic fat accumulation.
-
Insulin resistance: Genetic and pharmacological interventions that exacerbate or alleviate insulin resistance correspondingly increase or reduce hepatic steatosis and progression to MASH.
-
Mitochondrial dysfunction and oxidative stress: Restoration of mitochondrial function or antioxidant capacity attenuates hepatocellular injury, inflammation, and fibrogenesis.
-
Inflammatory and fibrogenic activation: Inhibition of Kupffer cell activation, hepatic stellate cell activation, or TGF-β signaling reduces fibrosis progression in multiple models.
Collectively, these findings support the essential role of each KE in driving the downstream sequence leading to adverse liver outcomes.
Evidence Assessment
Across the KERs in this AOP:
-
Biological plausibility is strong, grounded in established metabolic and inflammatory signaling pathways.
-
Empirical support is moderate to strong, with consistent directional responses observed across species and experimental systems.
-
Quantitative understanding is emerging, particularly for relationships linking insulin resistance, mitochondrial dysfunction, and oxidative stress, though gaps remain in defining precise thresholds and response-response relationships.
Overall, the weight of evidence supports a coherent and credible AOP suitable for regulatory use.
Known Modulating Factors
| Modulating Factor (MF) | Influence or Outcome | KER(s) involved |
|---|---|---|
| Nutritional status (e.g., high-fat diet) | Exacerbates insulin resistance and hepatic lipotoxicity | GR signaling → insulin resistance; FFA influx → steatosis |
| Sex hormones | Modulate GR sensitivity and metabolic responses | GR signaling → insulin resistance |
| Genetic susceptibility | Alters mitochondrial resilience and fibrotic responses | Mitochondrial dysfunction → oxidative stress; fibrosis progression |
| Inflammatory milieu | Amplifies cell injury and fibrogenesis | Oxidative stress → inflammation → fibrosis |
Quantitative Understanding
Quantitative data are available for several individual KERs (e.g., insulin resistance–steatosis, mitochondrial dysfunction–oxidative stress), but an integrated quantitative AOP model is not yet established. This AOP is therefore best applied in a qualitative or semi-quantitative context to support screening and prioritization rather than definitive risk characterization.
Considerations for Potential Applications of the AOP (optional)
This AOP may support:
-
Screening and prioritization of GR-modulating chemicals as potential MASLD contributors
-
Development of integrated testing strategies combining endocrine, metabolic, and hepatic endpoints
-
Grouping and read-across of EDCs with shared GR-mediated modes of action
-
Identification of high-value key events for assay development and refinement