
This AOP is licensed under the BY-SA license. This license allows reusers to distribute, remix, adapt, and build upon the material in any medium or format, so long as attribution is given to the creator. The license allows for commercial use. If you remix, adapt, or build upon the material, you must license the modified material under identical terms.
AOP: 638
Title
Co-exposure to microplastics and cadmium leading to progression from NAFLD to liver tumorigenesis
Short name
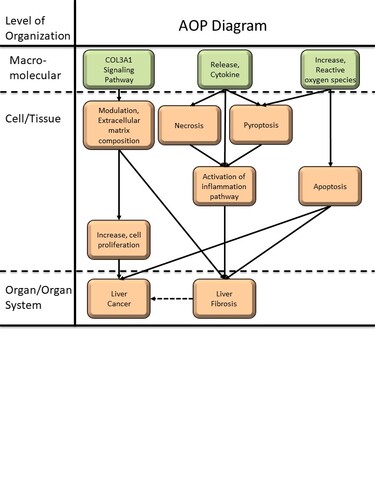
Graphical Representation
Point of Contact
Contributors
- Li Yuhan
- Wei Mu
Coaches
OECD Information Table
| OECD Project # | OECD Status | Reviewer's Reports | Journal-format Article | OECD iLibrary Published Version |
|---|---|---|---|---|
This AOP was last modified on March 29, 2026 02:53
Revision dates for related pages
| Page | Revision Date/Time |
|---|---|
| COL3A1 Signaling Pathway | March 25, 2026 08:17 |
| Release, Cytokine | September 16, 2017 10:14 |
| Increase, Reactive oxygen species | June 12, 2025 01:27 |
| modulation, Extracellular Matrix Composition | September 16, 2017 10:17 |
| Necrosis | February 07, 2017 13:22 |
| Pyroptosis | January 19, 2022 05:03 |
| Apoptosis | May 31, 2025 08:50 |
| Activation of inflammation pathway | May 31, 2022 02:47 |
| Increase, Cell Proliferation | December 10, 2024 15:01 |
| Increase, Liver fibrosis | February 11, 2026 05:35 |
| Liver Cancer | June 19, 2021 15:00 |
| COL3A1 Signaling Pathway leads to modulation, Extracellular Matrix Composition | March 25, 2026 08:36 |
| modulation, Extracellular Matrix Composition leads to Increase, Cell Proliferation | March 25, 2026 08:36 |
| Increase, Cell Proliferation leads to Liver Cancer | March 25, 2026 08:36 |
| Release, Cytokine leads to Necrosis | March 25, 2026 08:39 |
| Release, Cytokine leads to Apoptosis | March 25, 2026 08:39 |
| Release, Cytokine leads to Activation, inflammation pathway | March 25, 2026 08:41 |
| Necrosis leads to Activation, inflammation pathway | March 25, 2026 08:41 |
| Pyroptosis leads to Activation, inflammation pathway | March 25, 2026 08:42 |
| Activation, inflammation pathway leads to Increase, Liver fibrosis | March 25, 2026 08:49 |
| modulation, Extracellular Matrix Composition leads to Increase, Liver fibrosis | March 25, 2026 08:51 |
| Increase, ROS leads to Pyroptosis | March 25, 2026 08:51 |
| Increase, ROS leads to Apoptosis | April 11, 2024 15:22 |
| Apoptosis leads to Increase, Liver fibrosis | March 25, 2026 08:52 |
| Apoptosis leads to Liver Cancer | March 25, 2026 08:52 |
| Cadmium | October 25, 2017 08:33 |
| Nanoparticles and Micrometer Particles | February 04, 2022 13:43 |
Abstract
This Adverse Outcome Pathway (AOP) describes the pathological transformation of the liver from non-alcoholic fatty liver disease (NAFLD) to cirrhosis and ultimately to hepatocellular carcinoma under the combined exposure of microplastics and cadmium. The Molecular Initiating Events (MIEs) are characterized by COL3A1 signaling pathway, ROS generation, and cytokine aggregation. These triggers lead to a series of Key Events (KEs), including alterations in ECM organization, increased cell proliferation, the occurrence of three distinct types of cell death, and chronic inflammation. This biological cascade eventually results in two primary Adverse Outcomes (AOs): liver cancer and hepatic fibrosis. By delineating these relationships, this AOP provides a novel scientific perspective on the hepatotoxicity induced by the interaction between microplastics and heavy metals.
AOP Development Strategy
Context
Microplastics (MPs), fragments and particles ≤5 mm in diameter, persistently accumulate in the environment and have been detected in various human tissues and organs, including infant placental tissues. Once ingested via food or water, MPs can deposit in the liver, leading to significant tissue injury. Polystyrene microplastics (PS-MPs), widely used in disposable food packaging, are frequently employed as models to study these environmental effects. Beyond their inherent toxicity, PS-MPs can enter the human body and serve as carriers for other environmental pollutants. Among these co-exposed contaminants, heavy metals such as cadmium (Cd) are of particular concern due to their high carcinogenicity and shared exposure pathways with MPs through aquatic products, agricultural goods, and water resources.
Both MPs and Cd are ubiquitous pollutants that accumulate in the liver through food chain transfer and biomagnification, with absorbed dietary Cd exhibiting an exceptionally long biological half-life. As a primary target organ for both stressors, the liver undergoes amplified toxicological outcomes during co-exposure. Individual exposure to either Cd or MPs is known to induce oxidative stress, provoke inflammatory cascades, initiate apoptotic pathways, and disrupt hepatic metabolic homeostasis. When combined, these pollutants may result in a synergistic effect that accelerates pathological progression.
External stimuli and a sustained inflammatory microenvironment are key drivers in the development of liver cancer. This tumorigenic process is highly heterogeneous and involves a continuum of metabolic disorders, where Non-Alcoholic Fatty Liver Disease (NAFLD) serves as a significant precursor to liver fibrosis and eventual malignancy. Research indicates that long-term Cd exposure can drive the entire disease progression from NAFLD to fibrosis and liver cancer through the activation of endogenous reactive oxygen species (ROS). Furthermore, higher concentrations of plastic particles have been detected in cirrhotic liver tissues compared to healthy ones, and joint exposure to PS-MPs and Cd has been shown to promote fibrosis by regulating oxidative stress and cell death. Given that the transformation from inflammation to cancer involves complex molecular cascades and extended periods, this AOP is developed as a predictive tool to construct a hazard assessment framework for the synergistic hepatotoxicity of microplastics and heavy metals.
Strategy
Initial data on chemicals, genes, and disease interactions were collected from the CTD and GeneCards databases, focusing on the progression from NAFLD to liver cirrhosis and liver cancer. These target genes were categorized into three developmental stages and analyzed using GO and KEGG pathways, while protein-protein interaction networks were constructed to establish the molecular foundation. Following this preliminary predictive modeling, in vivo and in vitro experiments were conducted to provide empirical evidence for the proposed biological transitions.
Existing knowledge from the AOP-Wiki regarding cadmium and microplastics was further integrated to build a baseline network of pollutant-induced liver injury. By synthesizing enrichment analysis, interaction networks, and experimental results, the upstream and downstream relationships of key genes and pathways were identified and weighted. This approach, combining computational prediction and experimental verification, led to the final construction of the AOP mapping the progression from NAFLD through liver cirrhosis to liver cancer under combined exposure.
Summary of the AOP
Events:
Molecular Initiating Events (MIE)
Key Events (KE)
Adverse Outcomes (AO)
| Type | Event ID | Title | Short name |
|---|
| MIE | 2414 | COL3A1 Signaling Pathway | COL3A1 Signaling Pathway |
| MIE | 87 | Release, Cytokine | Release, Cytokine |
| MIE | 1115 | Increase, Reactive oxygen species | Increase, ROS |
| KE | 1195 | modulation, Extracellular Matrix Composition | modulation, Extracellular Matrix Composition |
| KE | 1263 | Necrosis | Necrosis |
| KE | 1967 | Pyroptosis | Pyroptosis |
| KE | 1262 | Apoptosis | Apoptosis |
| KE | 2009 | Activation of inflammation pathway | Activation, inflammation pathway |
| KE | 870 | Increase, Cell Proliferation | Increase, Cell Proliferation |
| AO | 344 | Increase, Liver fibrosis | Increase, Liver fibrosis |
| AO | 1395 | Liver Cancer | Liver Cancer |
Relationships Between Two Key Events (Including MIEs and AOs)
| Title | Adjacency | Evidence | Quantitative Understanding |
|---|
| COL3A1 Signaling Pathway leads to modulation, Extracellular Matrix Composition | adjacent | High | |
| modulation, Extracellular Matrix Composition leads to Increase, Cell Proliferation | adjacent | Moderate | |
| Increase, Cell Proliferation leads to Liver Cancer | adjacent | High | |
| Release, Cytokine leads to Necrosis | adjacent | High | |
| Release, Cytokine leads to Apoptosis | adjacent | High | |
| Release, Cytokine leads to Activation, inflammation pathway | adjacent | High | |
| Necrosis leads to Activation, inflammation pathway | adjacent | High | |
| Pyroptosis leads to Activation, inflammation pathway | adjacent | High | |
| Activation, inflammation pathway leads to Increase, Liver fibrosis | adjacent | High | |
| modulation, Extracellular Matrix Composition leads to Increase, Liver fibrosis | adjacent | High | |
| Increase, ROS leads to Pyroptosis | adjacent | High | |
| Increase, ROS leads to Apoptosis | adjacent | High | |
| Apoptosis leads to Increase, Liver fibrosis | adjacent | High | |
| Apoptosis leads to Liver Cancer | adjacent | Moderate |
Network View
Prototypical Stressors
Life Stage Applicability
| Life stage | Evidence |
|---|---|
| Not Otherwise Specified |
Taxonomic Applicability
| Term | Scientific Term | Evidence | Link |
|---|---|---|---|
| mammals | mammals | Moderate | NCBI |
Sex Applicability
| Sex | Evidence |
|---|---|
| Unspecific |
Overall Assessment of the AOP
By integrating bioinformatic analysis and findings from in vivo and in vitro experiments with established AOPs of pollutant-induced hepatic injury, a comprehensive AOP network is constructed to delineate the "inflammation-to-cancer" disease progression under the combined exposure to MPs and Cd.
Domain of Applicability
Molecular associations were derived from CTD and GeneCards to reflect environmental impacts on human health. Empirical validation was conducted in vivo using four-week-old male BALB/c mice, and in vitro using murine AML12 hepatocytes and human HepG2 liver cancer cells.
Essentiality of the Key Events
MIEs: COL3A1 signaling pathway; Release, cytokine (MIE: 87); and Increased, ROS (MIE: 1115) The essentiality of these MIEs is supported by the fact that increased reactive oxygen species and cytokine release govern the entire disease trajectory, acting as the indispensable molecular triggers for downstream pathological remodelling.
KE1: Modulation, Extracellular Matrix Composition (KE ID: 1195) As a structural protein essential to the ECM, COL3A1 expression is a requirement for ECM remodeling. The modulation of ECM composition is an essential functional transition leading to the adverse outcome of liver fibrosis.
KE2-4: Pyroptosis (KE ID: 1967); Necrosis (KE ID: 1263); and Apoptosis (KE ID: 1262) The occurrence of multiple programmed cell death pathways is critical for the "inflammation-to-cancer" transformation. These KEs are essential, as they serve as the bridge linking early oxidative stress and cytokine signaling to chronic tissue damage.
KE5: Activation of Inflammation (KE ID: 2009) Inflammatory activation is a necessary driver of the robust immune response and subsequent tissue damage. This KE is mediated by various forms of cell death and cytokines that occur across all disease stages.
KE6: Increase, Cell Proliferation (KE ID: 870) Cell proliferation is a fundamental requirement for tumorigenesis following chronic injury. It represents the necessary final step linking chronic inflammatory damage and ECM modulation to the development of liver cancer.
Evidence Assessment
MIEs to Cell Death and Cytokine Release Research indicates that combined exposure to microplastics and Cd significantly increases ROS levels and disrupts mitochondrial homeostasis, directly triggering the release of pro-inflammatory cytokines and initiating programmed cell death pathways.
MIEs to KE1 (ECM Modulation) PPI analysis reveals that COL3A1 signaling is a central node impacting both "inflammatory/NAFLD" and "liver cirrhosis" phases. In co-exposure models, COL3A1 is consistently enriched in ECM-related pathways, and the elevation of integrin ITGB5 further confirms the role of cell-ECM interactions.
KE2-4 (Cell Death) to KE5 (Inflammation) Analysis shows that necrosis-associated genes (BIRC3, RIPK3) and pyroptosis markers (CASP8) are enriched during disease progression. Necrosis and pyroptosis lead to cellular rupture and the release of intracellular contents, which activate the strong inflammatory response observed in animal models.
KE1 & KE5 to KE6 (Cell Proliferation) Bioinformatic evidence demonstrates that cell proliferation pathways are activated following ECM modulation and NF-κB-mediated inflammation. The ECM regulates cellular activities, while persistent inflammation stimulates the growth of initiated cells.
Pathway to Adverse Outcomes The integrated relationships among target genes and pathways facilitate the transition from NAFLD to liver fibrosis (defined by excessive ECM deposition) and ultimately to liver cancer (induced by sustained cell proliferation and inflammation).
Known Modulating Factors
| Modulating Factor (MF) | Influence or Outcome | KER(s) involved |
|---|---|---|
Quantitative Understanding
Considerations for Potential Applications of the AOP (optional)
References
[1] Li Y, Jian Y, Zhou J, Zhang M, Zhou Y, Ge Y, Wang H, Mu W. Molecular regulatory networks of microplastics and cadmium mediated hepatotoxicity from NAFLD to tumorigenesis via integrated approaches. Ecotoxicol Environ Saf. 2025 Jul 15;300:118431. doi: 10.1016/j.ecoenv.2025.118431.
[2] Wang, P., Chen, H., Kopittke, P. M., & Zhao, F.-J. (2019). Cadmium contamination in agricultural soils of China and the impact on food safety. Environmental Pollution, 249, 1038-1048. doi:10.1016/j.envpol.2019.03.063
[3] Wang, Y., Nan, X., Sun, H., Shi, Y., Miao, J., Li, Y., . . . Liu, B. (2024). From insects to mammals! Tissue accumulation and transgenerational transfer of micro/nano-plastics through the food chain. Journal of Hazardous Materials, 480. doi:10.1016/j.jhazmat.2024.136424
[4] Haouem, S., & El Hani, A. (2013). Effect of Cadmium on Lipid Peroxidation and on Some Antioxidants in the Liver, Kidneys and Testes of Rats Given Diet Containing Cadmium-polluted Radish Bulbs. Journal of Toxicologic Pathology, 26(4), 359-364. doi:10.1293/tox.2013-0025
[5] Horvatits, T., Tamminga, M., Liu, B., Sebode, M., Carambia, A., Fischer, L., . . . Fischer, E. K. (2022). Microplastics detected in cirrhotic liver tissue. eBioMedicine, 82. doi:10.1016/j.ebiom.2022.104147
[6] Lan, Y., Hu, L., Feng, X., Wang, M., Yuan, H., & Xu, H. (2024). Synergistic effect of PS-MPs and Cd on male reproductive toxicity: Ferroptosis via Keap1-Nrf2 pathway. Journal of Hazardous Materials, 461. doi:10.1016/j.jhazmat.2023.132584
[7] Li, J., Yin, K., Hou, L., Zhang, Y., Lu, H., Ma, C., & Xing, M. (2023). Polystyrene microplastics mediate inflammatory responses in the chicken thymus by Nrf2/NF-κB pathway and trigger autophagy and apoptosis. Environmental Toxicology and Pharmacology, 100. doi:10.1016/j.etap.2023.104136
[8] Sun, X.-H., Lv, M.-W., Zhao, Y.-X., Zhang, H., Ullah Saleem, M. A., Zhao, Y., & Li, J.-L. (2022). Nano-Selenium Antagonized Cadmium-Induced Liver Fibrosis in Chicken. Journal of Agricultural and Food Chemistry, 71(1), 846-856. doi:10.1021/acs.jafc.2c06562
[9] Sheng, S., Han, N., Wei, Y., Wang, J., Han, W., Xing, B., . . . Zhang, W. (2023). Liver Injury Induced by Exposure to Polystyrene Microplastics Alone or in Combination with Cadmium in Mice Is Mediated by Oxidative Stress and Apoptosis. Biological Trace Element Research, 202(5), 2170-2183. doi:10.1007/s12011-023-03835-5
[10] Wei, W., Yang, Q., Xiang, D., Chen, X., Wen, Z., Wang, X., . . . Xu, J. (2023). Combined impacts of microplastics and cadmium on the liver function, immune response, and intestinal microbiota of crucian carp (Carassius carassius). Ecotoxicology and Environmental Safety, 261. doi:10.1016/j.ecoenv.2023.115104
[11] Cichoz-Lach, H., & Michalak, A. (2014). Oxidative stress as a crucial factor in liver diseases. World Journal of Gastroenterology, 20(25), 8082-8091. doi:10.3748/wjg.v20.i25.8082
[12] Fontes-Cal, T. C. M., Mattos, R. T., Medeiros, N. I., Pinto, B. F., Belchior-Bezerra, M., Roque-Souza, B., . . . Gomes, J. A. S. (2021). Crosstalk Between Plasma Cytokines, Inflammation, and Liver Damage as a New Strategy to Monitoring NAFLD Progression. Frontiers in Immunology, 12. doi:10.3389/fimmu.2021.708959
[13] Tang, D., Kang, R., Berghe, T. V., Vandenabeele, P., & Kroemer, G. (2019). The molecular machinery of regulated cell death. Cell Research, 29(5), 347-364. doi:10.1038/s41422-019-0164-5
[14] Wu, Y., Zhang, J., Yu, S., Li, Y., Zhu, J., Zhang, K., & Zhang, R. (2022). Cell pyroptosis in health and inflammatory diseases. Cell Death Discovery, 8(1). doi:10.1038/s41420-022-00998-3