
This AOP is licensed under the BY-SA license. This license allows reusers to distribute, remix, adapt, and build upon the material in any medium or format, so long as attribution is given to the creator. The license allows for commercial use. If you remix, adapt, or build upon the material, you must license the modified material under identical terms.
AOP: 642
Title
Intestinal FXR inhibition leading to steatohepatitis via gut‑liver axis dysregulation
Short name
Graphical Representation
Point of Contact
Contributors
- Jung-Hwa Oh
Coaches
OECD Information Table
| OECD Project # | OECD Status | Reviewer's Reports | Journal-format Article | OECD iLibrary Published Version |
|---|---|---|---|---|
This AOP was last modified on April 28, 2026 04:06
Revision dates for related pages
| Page | Revision Date/Time |
|---|---|
| Inhibition, Intestinal FXR | April 22, 2026 21:50 |
| Ileal FGF15/FGF19 secretion, decreased | April 22, 2026 21:57 |
| Hepatic CYP7A1, increased | April 22, 2026 22:26 |
| Intrahepatic bile acid burden, increased | April 23, 2026 22:39 |
| Hepatic BSEP (ABCB11), decreased | April 23, 2026 22:42 |
| Bile acid composition in bile and intestine, altered | April 23, 2026 22:43 |
| Intestinal barrier, disruption | April 27, 2025 13:31 |
| Gut derived PAMPs, increased | April 23, 2026 22:47 |
| Increase, Oxidative Stress | February 11, 2026 07:05 |
| Inflammation, Liver | September 16, 2017 10:16 |
| Increase, Steatohepatitis | February 24, 2026 09:13 |
| FXR inhibition leads to Decreased FGF15/FGF19 | April 23, 2026 00:56 |
| Inflammation, Liver leads to Disruption of the intestinal barrier | April 27, 2026 21:19 |
| Decreased FGF15/FGF19 leads to Hepatic CYP7A1, increased | April 27, 2026 01:41 |
| Hepatic CYP7A1, increased leads to Intrahepatic bile acids, increased | April 27, 2026 01:42 |
| Intrahepatic bile acids, increased leads to BSEP (ABCB11), decreased | April 27, 2026 01:43 |
| BSEP (ABCB11), decreased leads to Bile acid composition, altered | April 27, 2026 01:44 |
| Bile acid composition, altered leads to Disruption of the intestinal barrier | April 27, 2026 01:44 |
| Disruption of the intestinal barrier leads to PAMPs, increased | April 27, 2026 01:44 |
| PAMPs, increased leads to Increase, Oxidative Stress | April 27, 2026 21:17 |
| Increase, Oxidative Stress leads to Inflammation, Liver | April 27, 2026 21:18 |
| Inflammation, Liver leads to Increase, Steatohepatitis | April 27, 2026 21:19 |
Abstract
Steatohepatitis is the inflammatory form of steatotic liver disease and a major driver of progressive liver damage, fibrosis and hepatocellular carcinoma. The gut‑liver axis, and particularly bile acid–farnesoid X receptor (FXR) signalling, plays a central role in the development of steatohepatitis, but an AOP formalising the causal sequence from intestinal FXR inhibition to steatohepatitis has not been available.
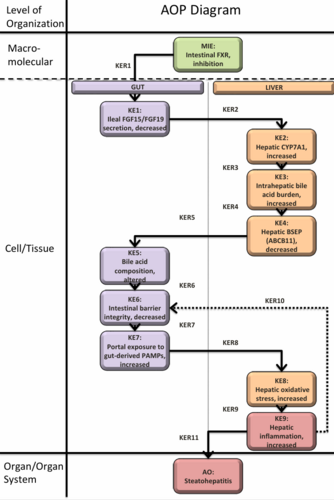
This AOP describes how inhibition of intestinal FXR (MIE) can culminate in steatohepatitis (AO) through a linear sequence of well‑defined key events that integrate endocrine, metabolic, and barrier‑immune mechanisms. Inhibition of ileal FXR reduces FGF15/FGF19 secretion (KE1), derepressing hepatic CYP7A1 (KE2) and thereby increasing bile acid synthesis and intrahepatic bile acid burden (KE3). Under these conditions, bile salt export pump (BSEP/ABCB11) expression and function are reduced (KE4), contributing together with excess synthesis to quantitative and qualitative alterations of bile acid composition in bile and along the intestine (KE5). The resulting enrichment of hydrophobic and microbially derived bile acids, combined with loss of FXR‑mediated epithelial defence, impairs intestinal barrier integrity (KE6) and increases portal exposure to gut‑derived pathogen‑associated molecular patterns (PAMPs) such as lipopolysaccharide (KE7). On the hepatic side, bile acid overload and lipid accumulation promote oxidative stress (KE8), while PAMP‑ and damage‑associated signalling activate Kupffer cells and other immune cells, driving hepatic inflammation (KE9) that, together with steatosis, manifests histologically as steatohepatitis (AO).
The biological plausibility of this AOP is high, as each KER follows established physiology of the FXR–FGF15/FGF19 axis, enterohepatic bile acid circulation, gut barrier biology, and inflammatory signalling in the liver. Essentiality of the central KEs is supported by intestinal and hepatic FXR knockout models, genetic and pharmacologic manipulation of FGF15/FGF19 and CYP7A1, experimental modulation of bile acid composition and intestinal permeability, and interventions that alter portal PAMP load, oxidative stress, and hepatic inflammation in rodent NASH and cholestasis models. Clinical and translational data showing dysregulation of FXR–FGF19 signalling, bile acid profiles, and gut–liver barrier function in patients with steatotic liver disease and steatohepatitis further support human relevance. This AOP is therefore suited to underpin assay development and integrated approaches to testing and assessment (IATA) for chemicals or drugs that perturb intestinal FXR and bile acid signalling, to help prioritise potential steatogenic or steatohepatitic liabilities and to provide a mechanistic context for regulatory interpretation of gut–liver axis–mediated liver injury.
AOP Development Strategy
Context
Steatohepatitis develops in the setting of steatotic liver disease when lipid overload is accompanied by hepatocellular injury and inflammation. Over the past decade, research has highlighted that this transition is strongly influenced by the gut–liver axis rather than by hepatic mechanisms alone. Intestinal FXR, highly expressed in the ileum, is a central regulator of bile acid composition, FGF15/FGF19 secretion and intestinal barrier function, all of which shape hepatic exposure to bile acids and microbial products. Dysregulation of this signalling axis has been repeatedly associated with steatosis, steatohepatitis and fibrosis in experimental models and in humans. This AOP focuses specifically on perturbations of intestinal FXR within the gut–liver axis as a biologically plausible and translationally relevant driver of steatohepatitis, with the aim of providing a structured mechanistic framework that can support assay development, IATA design and regulatory interpretation, without attempting to cover all possible causes or pathways leading to this liver outcome.
Strategy
The development of this AOP was guided by a targeted, mechanism‑oriented literature strategy focused on three elements: (1) organ‑specific roles of FXR, with emphasis on intestinal FXR; (2) the FXR–FGF15/FGF19–CYP7A1 feedback loop and bile acid homeostasis; and (3) gut–liver axis processes linking bile acids, intestinal barrier function, microbial products and steatohepatitis. Initial scoping relied on expert knowledge and key reviews to propose the MIE, candidate KEs and a provisional linear sequence from intestinal FXR inhibition to steatohepatitis.
Subsequent searches in major biomedical databases used combinations of terms such as “intestinal FXR”, “FGF15/FGF19”, “CYP7A1”, “bile acid pool/composition”, “BSEP/ABCB11”, “intestinal permeability”, “endotoxin/LPS”, “gut–liver axis” and “NASH/steatohepatitis”, together with “knockout”, “overexpression”, “agonist” or “inhibitor” to prioritise mechanistic and intervention studies. Titles/abstracts were first screened to retain experimental animal, in vitro and translational clinical studies with mechanistic endpoints, followed by full‑text review to identify direct gain‑ and loss‑of‑function evidence for each KE and KER, as well as consistent correlative data across models and species. The final AOP scope was intentionally limited to KEs and KERs with at least moderate support for biological plausibility, empirical linkage and essentiality in the context of intestinal FXR perturbation and steatohepatitis, in order to maximise clarity and reusability for regulatory applications.
Summary of the AOP
Events:
Molecular Initiating Events (MIE)
Key Events (KE)
Adverse Outcomes (AO)
| Type | Event ID | Title | Short name |
|---|
| MIE | 2421 | Inhibition, Intestinal FXR | FXR inhibition |
| KE | 2422 | Ileal FGF15/FGF19 secretion, decreased | Decreased FGF15/FGF19 |
| KE | 2424 | Hepatic CYP7A1, increased | Hepatic CYP7A1, increased |
| KE | 2425 | Intrahepatic bile acid burden, increased | Intrahepatic bile acids, increased |
| KE | 2426 | Hepatic BSEP (ABCB11), decreased | BSEP (ABCB11), decreased |
| KE | 2427 | Bile acid composition in bile and intestine, altered | Bile acid composition, altered |
| KE | 1931 | Intestinal barrier, disruption | Disruption of the intestinal barrier |
| KE | 2428 | Gut derived PAMPs, increased | PAMPs, increased |
| KE | 1392 | Increase, Oxidative Stress | Increase, Oxidative Stress |
| KE | 902 | Inflammation, Liver | Inflammation, Liver |
| AO | 1489 | Increase, Steatohepatitis | Increase, Steatohepatitis |
Relationships Between Two Key Events (Including MIEs and AOs)
| Title | Adjacency | Evidence | Quantitative Understanding |
|---|
| FXR inhibition leads to Decreased FGF15/FGF19 | adjacent | High | Moderate |
| Decreased FGF15/FGF19 leads to Hepatic CYP7A1, increased | adjacent | High | Moderate |
| Hepatic CYP7A1, increased leads to Intrahepatic bile acids, increased | adjacent | High | Moderate |
| Intrahepatic bile acids, increased leads to BSEP (ABCB11), decreased | adjacent | Moderate | Low |
| BSEP (ABCB11), decreased leads to Bile acid composition, altered | adjacent | Moderate | Low |
| Bile acid composition, altered leads to Disruption of the intestinal barrier | adjacent | Moderate | Low |
| Disruption of the intestinal barrier leads to PAMPs, increased | adjacent | High | Low |
| PAMPs, increased leads to Increase, Oxidative Stress | adjacent | Moderate | Low |
| Increase, Oxidative Stress leads to Inflammation, Liver | adjacent | Moderate | Low |
| Inflammation, Liver leads to Increase, Steatohepatitis | adjacent | High | Moderate |
| Inflammation, Liver leads to Disruption of the intestinal barrier | non-adjacent | Moderate | Low |
Network View
Prototypical Stressors
Life Stage Applicability
Taxonomic Applicability
Sex Applicability
Overall Assessment of the AOP
This AOP is primarily applicable to mammals with a conserved FXR–FGF15/FGF19–CYP7A1 axis and enterohepatic bile acid circulation, notably rodents and humans. It is most strongly supported for adult life stages; while both sexes are represented in the underlying studies, sex‑specific differences in FXR and bile acid metabolism are not explicitly resolved and are therefore considered within the general adult domain of applicability.
The overall weight of evidence is moderate‑to‑high. Biological plausibility of the key event sequence from intestinal FXR inhibition through altered bile acid synthesis and composition, intestinal barrier dysfunction, increased portal PAMP exposure, hepatic oxidative stress and inflammation to steatohepatitis is strong and aligned with current understanding of gut–liver axis biology. Empirical support for several central KERs (FXR–FGF15/FGF19–CYP7A1–bile acid burden, bile acid–barrier interactions, barrier/PAMP–driven hepatic inflammation) includes both genetic and pharmacologic gain‑ and loss‑of‑function data, as well as dose‑ and time‑dependent concordance in multiple rodent models.
On this basis, the AOP is suitable as a qualitative mechanistic framework for regulatory uses such as early hazard flagging and prioritisation of intestinal FXR‑modulating substances, design and interpretation of gut–liver axis testing strategies, and weight‑of‑evidence support in risk assessment where steatohepatitis is a concern. Quantitative relationships and explicit modulating factors (diet, microbiota, genetics, sex) are not yet fully defined, so use in strictly quantitative prediction should be considered with caution.
Domain of Applicability
This AOP is intended to apply to species, life stages and biological contexts in which the intestinal FXR–FGF15/FGF19–CYP7A1 axis and enterohepatic bile acid circulation are conserved and functionally important.
Taxa
-
The primary taxonomic domain is mammals, especially rodents (mouse, rat) and humans, where FXR, FGF15/FGF19, FGFR4/β‑Klotho, CYP7A1, BSEP and gut–liver barrier–immune mechanisms are well described.
-
Applicability to non‑mammalian vertebrates (e.g. fish, birds) is uncertain, because of species differences in bile acid chemistry, FXR ligands, and the presence and function of FGF15/19 and enterohepatic circulation. At present, the AOP should not be extrapolated beyond mammals without additional species‑specific data.
Life stage
-
Evidence underpinning the AOP comes predominantly from adult animals and adult human patients with steatotic liver disease or cholestatic conditions.
-
Data in juvenile or aged individuals are more limited, and developmental changes in bile acid metabolism and intestinal barrier function may alter sensitivity and kinetics. Consequently, the AOP is most robustly supported for adolescent to adult life stages.
Sex
-
Both males and females are represented in the experimental and clinical literature that informs this AOP, and the core FXR–FGF15/FGF19–CYP7A1 axis and gut–liver barrier mechanisms are conserved between sexes.
-
Known sex differences in bile acid pool size, composition and FXR activity may influence quantitative responses and susceptibility, but do not invalidate the qualitative sequence of key events. Thus, the AOP is considered applicable to both sexes, while recognising that sex may act as a modulating factor.
Other biological context
-
The AOP is most relevant under conditions where enterohepatic bile acid cycling is intact and the intestinal barrier and microbiota are present (i.e. conventional, non‑germ‑free animals and humans).
-
Dietary factors (e.g. high‑fat/high‑sugar diets), metabolic status (obesity, insulin resistance) and concomitant gut microbiota changes can modulate the sensitivity and magnitude of the key events, but the qualitative event sequence is expected to hold across a range of such backgrounds.
Essentiality of the Key Events
- MIE – Intestinal FXR, inhibition
- Essentiality: Moderate (mainly indirect, pathway‑level evidence)
Intestine‑specific or whole‑body FXR‑deficient mice show reduced ileal FGF15/FGF19 expression, increased hepatic CYP7A1 and bile acid synthesis, expansion of the bile acid pool, and aggravated steatosis, inflammation and fibrosis under dietary or toxic challenges compared with wild‑type controls, indicating that loss of intestinal FXR signalling worsens multiple downstream KEs and the AO. Conversely, pharmacological activation of intestinal FXR with gut‑restricted or gut‑biased agonists increases ileal FGF15/FGF19, suppresses CYP7A1, normalises bile acid synthesis and attenuates hepatic steatosis and inflammation in NASH models. Because most interventions modulate both intestinal and hepatic FXR and other pathways simultaneously, the essentiality of intestinal FXR inhibition per se is supported primarily by pathway‑level, but consistent, evidence and is therefore graded as Moderate.
KE1 – Ileal FGF15/FGF19 secretion, decreased Essentiality: High (direct evidence)
Mice lacking intestinal FXR or Fgf15 exhibit markedly reduced ileal FGF15 expression, increased hepatic CYP7A1 and C4, elevated bile acid synthesis, enlarged bile acid pool and worsened steatosis and liver injury under dietary challenge compared with wild‑type animals, demonstrating that loss of KE1 enhances multiple downstream KEs. Conversely, restoring or enhancing FGF15/FGF19 signalling by FGF19 analogs, adenoviral FGF19 expression or FXR agonists suppresses CYP7A1, reduces bile acid synthesis and intrahepatic bile acid burden, and improves steatosis, inflammation and fibrosis endpoints in rodent models. These gain‑ and loss‑of‑function data directly show that blocking or increasing KE1 has the expected impact on downstream KEs and the AO, supporting a High essentiality rating.
- KE2 – Hepatic CYP7A1 expression/activity, increased
- Essentiality: High (direct evidence)
Transgenic overexpression of Cyp7a1 increases conversion of cholesterol to bile acids, expands the bile acid pool and alters bile acid composition, which is accompanied by changes in liver injury and metabolic phenotypes, whereas Cyp7a1 knockout mice display a reduced bile acid pool and decreased bile acid synthesis. In FXR/FGF15/FGF19‑manipulated models, suppression of CYP7A1 consistently lowers bile acid synthesis and intrahepatic bile acid burden and mitigates downstream injury, while failure to suppress CYP7A1 maintains or worsens these outcomes. Together, these specifically designed gain‑ and loss‑of‑function studies show that modulating KE2 directly controls downstream KEs related to bile acid burden and contributes to the AO, justifying a High essentiality rating.
- KE3 – Intrahepatic bile acid burden, increased
- Essentiality: High (direct evidence)
Experimental elevation of intrahepatic bile acid levels by bile acid loading, increased synthesis or impaired export produces cholestatic liver injury with hepatocellular damage, inflammation and fibrosis in animal models, demonstrating that increased KE3 can by itself drive downstream KEs. Conversely, reducing intrahepatic bile acid burden with bile acid sequestrants, FXR or FGF19 agonists, or inhibitors of bile acid synthesis decreases hepatic bile acid content and consistently attenuates oxidative stress, inflammation, fibrosis and steatohepatitis severity. These interventions directly link quantitative changes in KE3 to corresponding changes in multiple downstream KEs and the AO, supporting High essentiality.
- KE4 – Hepatic BSEP (ABCB11) expression/function, decreased
- Essentiality: Moderate (direct for cholestasis, indirect in this AOP)
Genetic BSEP deficiency (PFIC2), Bsep knockout models and pharmacologic BSEP inhibition cause impaired canalicular bile acid export, marked intrahepatic bile acid accumulation and cholestatic liver injury with inflammation and fibrosis, indicating that loss of KE4 can be sufficient to exacerbate downstream KEs related to bile acid burden and liver damage. Restoration or upregulation of BSEP expression and function, for example by FXR agonists or geniposide, improves bile flow, lowers hepatic bile acid levels and ameliorates markers of liver injury and fibrosis. However, in the specific context where bile acid overproduction and other perturbations co‑occur, BSEP is one of several contributors to KE3 and KE5 rather than a uniquely isolated node, so its essentiality in this linear AOP is best characterised as Moderate, supported by strong cholestasis data but more indirect evidence for the full gut–liver steatohepatitis sequence.
- KE5 – Bile acid composition in bile and intestine, altered
- Essentiality: Moderate (indirect but consistent evidence)
Experimental manipulation of bile acid composition—through genetic or pharmacologic modification of bile acid synthesis enzymes, intestinal transporters or microbial bile salt hydrolases—shifts the balance between primary and secondary, conjugated and unconjugated, and hydrophilic and hydrophobic bile acids, and these shifts are associated with parallel changes in intestinal barrier integrity, gut microbiota, hepatic inflammation and fibrosis. Interventions that maintain a more conjugated, less hydrophobic bile acid pool protect barrier function and reduce liver injury, whereas enrichment of toxic secondary or hydrophobic bile acids aggravates barrier dysfunction and hepatic inflammation. Because these studies typically modulate bile acid amount, composition and receptor signalling together, the essentiality of KE5 is well supported but largely indirect, and thus graded as Moderate.
- KE6 – Intestinal barrier integrity, decreased
- Essentiality: High (direct evidence)
In models of fatty liver disease and cholestatic liver injury, experimental disruption of the intestinal barrier (e.g. DSS colitis, MCD diet, other mucosal insults) increases intestinal permeability, portal endotoxin levels and hepatic inflammation and fibrosis compared with barrier‑intact controls, demonstrating that inducing KE6 exacerbates downstream KEs and the AO. Conversely, preserving or restoring barrier integrity using probiotics, prebiotics, antibiotics, FXR/TGR5/PXR agonists or tight junction‑stabilising agents reduces intestinal leakiness, lowers portal PAMP exposure and significantly attenuates hepatic inflammation and fibrotic progression. These specifically designed interventions clearly show that blocking or reversing KE6 reduces the magnitude of downstream KEs and the AO, supporting a High essentiality rating.
- KE7 – Portal exposure to gut-derived PAMPs (e.g. LPS), increased
- Essentiality: High (direct evidence)
Diet‑induced NASH, alcohol, MCD diet and colitis models consistently demonstrate that increased portal or systemic levels of LPS and other bacterial products are associated with stronger hepatic inflammatory responses and fibrosis compared with animals with lower PAMP exposure. Direct reduction of PAMP load through broad‑spectrum antibiotics, germ‑free conditions, TLR4/MyD88 knockout or LPS neutralisation diminishes hepatic inflammatory signalling, inflammatory infiltrates and fibrotic progression, even in the presence of persistent steatosis or metabolic stress. These data provide direct evidence that modulating KE7 in either direction has predictable effects on downstream KEs and the AO, supporting High essentiality.
- KE8 – Hepatic oxidative stress, increased
- Essentiality: Moderate (amplifier rather than absolute requirement)
Numerous NASH, cholestasis and toxin models show that hepatic oxidative stress markers (ROS, MDA, 4‑HNE, 8‑OHdG) increase in step with steatosis, inflammation and fibrosis, and that higher levels of oxidative stress correlate with more severe disease. Antioxidant therapies, Nrf2 activators, mitochondrial protectants and inhibitors of lipid peroxidation reduce oxidative stress and often attenuate hepatic inflammation and fibrotic progression, indicating that lowering KE8 can mitigate downstream KEs and the AO. Nevertheless, some degree of steatohepatitis can still develop in models where oxidative stress is partially controlled, reflecting parallel contributions from PAMP‑driven inflammation and direct bile acid toxicity, so KE8 is best viewed as an important amplifier with Moderate essentiality rather than an absolutely indispensable step.
- KE9 – Hepatic inflammation, increased
- Essentiality: High (direct evidence)
Across diet‑induced NASH, cholestatic liver injury and FXR/FGF19 dysregulation models, hepatic inflammation—measured by inflammatory infiltrates, cytokine and chemokine expression and histological inflammation scores—is closely associated with progression from simple steatosis to steatohepatitis and fibrosis. Interventions that directly suppress hepatic inflammatory signalling, including FXR agonists, bile acid modulators, TLR4/MyD88 inhibitors, anti‑TNF agents and other anti‑inflammatory therapies, reduce inflammatory markers and are frequently accompanied by improvements in ballooning, steatosis and fibrosis, and even regression of established steatohepatitis in some models. These studies show that attenuating KE9 consistently reduces downstream AO severity, supporting a High essentiality rating for hepatic inflammation in this AOP.
Evidence Assessment
- KER1: Intestinal FXR, inhibition → Ileal FGF15/FGF19 secretion, decreased
- Biological plausibility: Intestinal FXR directly binds FXR response elements in the FGF15/FGF19 promoter, and bile acid–induced FXR activation in the distal ileum is a well‑established driver of FGF15/FGF19 transcription and secretion. Loss or inhibition of intestinal FXR is therefore expected to reduce FGF15/FGF19 expression as a direct transcriptional consequence.
- Empirical support: Intestine‑specific and whole‑body FXR knockout mice show markedly reduced ileal Fgf15 and circulating FGF15/FGF19 under basal and bile acid–stimulated conditions, whereas pharmacologic FXR agonists robustly increase FGF15/FGF19 in rodents and humans. Time‑course and diurnal studies confirm that FXR activity and FGF15/FGF19 levels fluctuate in parallel. Overall, empirical support is strong and consistent.
- Quantitative understanding: Dose–response data show graded induction of ileal FGF15/FGF19 with increasing bile acid or FXR agonist exposure, but fully quantitative models linking a defined decrement in intestinal FXR activity to FGF15/FGF19 output across species and conditions are not yet established. Quantitative understanding is therefore considered Moderate.
- KER2: Ileal FGF15/FGF19 secretion, decreased → Hepatic CYP7A1 expression/activity, increased
- Biological plausibility: FGF15/FGF19 is the key endocrine mediator of negative feedback from intestine to liver, acting via FGFR4/β‑Klotho and downstream signalling (e.g. ERK/JNK/Src) to repress CYP7A1 transcription. Thus, reduced FGF15/FGF19 signalling is expected to derepress CYP7A1 and increase bile acid synthesis.
- Empirical support: Fgf15‑deficient or FGFR4/β‑Klotho‑deficient mice show increased hepatic Cyp7a1 expression, elevated C4 and higher bile acid synthesis, whereas exogenous FGF19 or FXR agonists that restore FGF19 signalling reduce CYP7A1 and bile acid synthesis in rodents and humans. Human studies show an inverse quantitative correlation between serum FGF19 and C4 (a CYP7A1 activity surrogate). Empirical support is strong and consistent across models.
- Quantitative understanding: Good quantitative relationships between FGF19 and C4 have been demonstrated in humans, and dose–response data for FGF19 analogues on CYP7A1/C4 exist in clinical and preclinical studies. However, integrated models spanning intestinal FXR perturbation → FGF15/FGF19 → hepatic CYP7A1 across species and contexts are incomplete; quantitative understanding is graded as Moderate.
- KER3: Hepatic CYP7A1 expression/activity, increased → Intrahepatic bile acid burden, increased
- Biological plausibility: CYP7A1 catalyses the rate‑limiting step in the classic pathway of bile acid synthesis from cholesterol, so increased CYP7A1 activity is expected to increase de novo bile acid production and, unless fully compensated by excretion, increase hepatic bile acid burden and pool size.
- Empirical support: Cyp7a1 transgenic overexpression in mice increases bile acid synthesis, expands the bile acid pool and alters hepatic and biliary bile acid levels, whereas Cyp7a1 knockout reduces pool size and synthesis. In FXR/FGF19‑manipulated models, higher CYP7A1 expression is consistently associated with higher intrahepatic bile acid content and vice versa. Empirical support is strong.
- Quantitative understanding: Transgenic and knockout studies provide semi‑quantitative relationships (e.g. approximately two‑fold increase in CYP7A1 activity leading to approximately 2–2.5‑fold expansion of the bile acid pool), but precise dose–response curves and human‑relevant quantitative functions are limited. Quantitative understanding is considered Moderate.
- KER4: Intrahepatic bile acid burden, increased → Hepatic BSEP (ABCB11) expression/function, decreased
- Biological plausibility: Under physiological conditions, bile acids activate FXR to induce BSEP; however, sustained bile acid overload combined with cholestatic and inflammatory stress can downregulate BSEP transcription and impair its localisation and function via ER stress, inflammatory cytokines (e.g. IL‑1β, TNF‑α) and altered FXR signalling. Thus, pathological increases in intrahepatic bile acid burden are plausibly linked to reduced effective BSEP function.
- Empirical support: Cholestatic models and drug‑induced liver injury show that intrahepatic bile acid accumulation is accompanied by decreased BSEP mRNA/protein, mislocalisation of BSEP at the canalicular membrane and worsened bile acid retention. Interventions that reduce bile acid burden or inflammation partially restore BSEP expression and function. However, many studies manipulate bile acids, inflammation and FXR together, making it difficult to isolate bile acid burden as the sole driver of BSEP changes. Empirical support is therefore Moderate and context‑dependent.
- Quantitative understanding: There are no well‑defined quantitative response–response functions linking a specific rise in intrahepatic bile acid levels to a defined decrement in BSEP expression/function across conditions. Quantitative understanding is Low–Moderate and best considered Low for AOP purposes.
- KER5: Intrahepatic bile acid burden / BSEP dysfunction → Bile acid composition in bile and intestine, altered
- Biological plausibility: Increased bile acid synthesis (via CYP7A1) and impaired canalicular export (via reduced BSEP) together modify the flux and routing of bile acids through the biliary tree and intestine, affecting the relative abundance of primary vs secondary, conjugated vs unconjugated and hydrophilic vs hydrophobic bile acids. These changes are further shaped by altered intestinal re‑uptake and microbial metabolism. It is therefore plausible that bile acid overproduction plus export dysfunction leads to altered bile acid composition in bile and the gut lumen.
- Empirical support: Cyp7a1 transgenic and cholestatic models exhibit both increased bile acid pool size and clear shifts in bile acid species (e.g. CA/CDCA ratio, secondary bile acid fractions), while BSEP deficiency/inhibition and FXR disruption also modify biliary and intestinal bile acid profiles. Studies combining modern bile acid profiling with genetic or pharmacologic manipulations document consistent compositional changes, although amount and composition are often co‑modulated. Empirical support is Moderate‑to‑strong but usually not specific to a single upstream KE.
- Quantitative understanding: High‑resolution analytical methods allow quantitative measurement of individual bile acids, but formal quantitative models predicting compositional shifts from a defined increase in CYP7A1 activity or bile acid burden are lacking. Quantitative understanding of this KER is Low–Moderate and best treated as Low in regulatory contexts.
- KER6: Bile acid composition in bile and intestine, altered → Intestinal barrier integrity, decreased
- Biological plausibility: Bile acids act both as detergents and signalling molecules. Enrichment of hydrophobic, deconjugated or secondary bile acids increases epithelial toxicity, tight junction disruption and mucosal inflammation, whereas more hydrophilic/conjugated bile acids can be barrier‑protective via FXR/TGR5 signalling. A shift towards more toxic bile acid profiles is therefore mechanistically expected to impair intestinal barrier integrity.
- Empirical support: In vivo, diets and interventions that increase colonic exposure to hydrophobic or secondary bile acids are associated with increased permeability and reduced tight junction proteins, while maintaining a more conjugated or less hydrophobic pool (e.g. by microbial BSH inhibition or specific bile acid supplementation) preserves or improves barrier function and reduces liver injury. In vitro, exposure of intestinal epithelial cells or organoids to higher concentrations of deoxycholic or lithocholic acid disrupts tight junctions and increases paracellular permeability, whereas more hydrophilic bile acids are less damaging and sometimes protective. Empirical support is Moderate‑to‑strong but often involves concomitant microbiota and receptor effects.
- Quantitative understanding: Some studies report concentration‑dependent barrier disruption for specific bile acids, but comprehensive quantitative functions linking complex in vivo composition changes to barrier integrity are not available. Quantitative understanding is Low–Moderate and for this AOP best classified as Low.
- KER7: Intestinal barrier integrity, decreased → Portal exposure to gut‑derived PAMPs, increased
- Biological plausibility: The intestinal barrier—epithelial cells, tight junctions and mucus—physically restricts luminal microbes and their products from entering the portal circulation. Barrier disruption increases paracellular and transcellular translocation of bacteria and PAMPs (e.g. LPS, peptidoglycan, flagellin), making an increase in portal PAMP exposure a direct and plausible consequence.
- Empirical support: In multiple models (DSS colitis, MCD diet, high‑fat diet, alcohol), increased intestinal permeability (measured by FITC‑dextran flux, zonulin elevation and tight junction loss) coincides with higher portal or systemic LPS and bacterial DNA levels. Barrier‑protective interventions reduce both permeability and portal endotoxin levels, while deliberate barrier injury increases PAMP translocation. Empirical support is strong and consistent.
- Quantitative understanding: Several studies show correlations between quantitative measures of permeability and portal endotoxin, but system‑level quantitative models are limited and often confounded by changes in microbiota, motility and bile flow. Quantitative understanding is Low–Moderate and best considered Low.
- KER8: Portal exposure to gut‑derived PAMPs, increased → Hepatic oxidative stress, increased
- Biological plausibility: Portal PAMPs (e.g. LPS, peptidoglycan, bacterial DNA) engage TLR4 and other pattern‑recognition receptors on Kupffer cells and hepatic endothelial cells, triggering NADPH oxidase (NOX2) activation, mitochondrial electron transport chain disruption and NF‑κB–mediated pro‑oxidant gene expression, all of which are well‑established mechanisms of ROS generation in the liver. Increased portal PAMP load is therefore mechanistically expected to elevate intrahepatic ROS, lipid peroxidation and oxidative DNA damage independently of, and in addition to, the contribution of bile acid overload and steatosis.
- Empirical support: Rodent models of endotoxaemia and gut‑derived PAMP exposure show elevated hepatic ROS, MDA, 4‑HNE and 8‑OHdG levels alongside Kupffer cell activation and inflammatory responses. TLR4/MyD88 knockout or LPS neutralisation reduces both oxidative stress markers and downstream hepatic inflammation, whereas direct LPS administration to steatotic animals increases hepatic ROS in a dose‑dependent manner. Targeted interventions using NADPH oxidase inhibitors (e.g. apocynin, DPI) or NOX2 KO models further show that blocking PAMP‑induced ROS generation can partially uncouple oxidative stress from downstream inflammation, providing mechanistically direct evidence for this KER. Overall, empirical support is Moderate.
- Quantitative understanding: Some dose–time‑course studies document graded increases in hepatic oxidative stress markers in response to defined LPS doses in rodents, but fully parameterised quantitative models linking portal PAMP concentration to specific ROS output across species and metabolic states are not yet available. Confounding by simultaneous inflammatory and metabolic changes further limits quantitative interpretation. Quantitative understanding is therefore Low.
- KER9: Hepatic oxidative stress, increased → Hepatic inflammation, increased
- Biological plausibility: ROS and lipid peroxidation products generate damage‑associated molecular patterns (DAMPs) that activate Kupffer cells and hepatic stellate cells via pattern‑recognition and stress‑response pathways, amplifying inflammatory cytokine production and recruiting immune cells. Oxidative stress is therefore a plausible proximal driver and amplifier of hepatic inflammation, acting in concert with PAMP‑driven and bile acid–driven immune activation.
- Empirical support: In many NASH and toxin models, increased oxidative stress co‑localises with areas of inflammatory infiltration, and higher ROS/oxidative damage is associated with stronger inflammatory responses. Antioxidant and Nrf2‑activating interventions that reduce oxidative stress often decrease inflammatory cytokine expression and inflammatory cell infiltration, though not always to baseline, indicating a contributory but not exclusive role. Empirical support is Moderate.
- Quantitative understanding: There is limited quantitative information on exact thresholds of oxidative stress required to trigger specific levels of hepatic inflammation, and multiple parallel pathways (e.g. PAMP‑driven signalling, direct bile acid toxicity) complicate modelling. Quantitative understanding is therefore Low.
- KER10: Hepatic inflammation, increased → Intestinal barrier integrity, decreased (gut–liver feedback)
- Biological plausibility: Hepatic inflammation releases pro‑inflammatory cytokines and altered bile acid mixtures into the systemic circulation and bile, which can impair epithelial turnover, disrupt tight junctions and disturb mucus and antimicrobial defences in the intestine. These effects, together with inflammation‑induced changes in bile acid composition and microbiota, provide a coherent mechanistic basis for liver inflammation to further weaken intestinal barrier integrity and reinforce gut–liver axis dysregulation.
- Empirical support: Patients with advanced chronic liver disease or severe steatohepatitis frequently show increased intestinal permeability that correlates with global liver dysfunction and systemic inflammation, and not solely with primary gut disease. In animal models, interventions that mainly reduce hepatic inflammation or cholestasis (e.g. FXR agonists, anti‑inflammatory or bile acid–modulating drugs) often improve barrier markers and microbiota composition, suggesting a liver‑to‑gut feedback effect, although most studies do not isolate hepatic inflammation as the only variable. Overall, empirical support is Moderate.
- Quantitative understanding: Available human and animal data consistently show that intestinal permeability tends to be higher in more advanced or inflamed liver disease, but quantitative response–response relationships between defined changes in hepatic inflammation and specific barrier endpoints are scarce and heterogeneous. This KER currently has Low quantitative understanding: the direction and plausibility are clear, but no robust predictive function can yet be defined.
- KER11: Hepatic inflammation, increased → Steatohepatitis (AO)
- Biological plausibility: Steatohepatitis is defined histologically by the co‑occurrence of hepatic steatosis, hepatocellular ballooning and lobular inflammation, with or without fibrosis; hepatic inflammation is therefore not merely associated with but definitionally required for the diagnosis of steatohepatitis. Sustained hepatic inflammatory signalling drives hepatocellular injury and ballooning via cytotoxic cytokines (e.g. TNF‑α, IL‑1β), activates hepatic stellate cells and promotes fibrogenesis, and perpetuates lipid accumulation by impairing fatty acid oxidation and lipid export, thereby completing the histological picture of steatohepatitis. Anti‑inflammatory interventions in NASH models consistently reduce NAS inflammation and ballooning sub‑scores together, confirming that inflammation is a direct upstream driver of the AO.
- Empirical support: In rodent NASH models (high‑fat diet, MCD diet, STAM, CDAHFD), the severity of hepatic inflammation as measured by inflammatory cell infiltration, cytokine levels and NAS inflammation scores closely tracks the development and severity of steatohepatitis and fibrosis. Genetic or pharmacologic suppression of hepatic inflammatory signalling—including FXR agonists, TLR4/MyD88 inhibition, anti‑TNF agents, NLRP3 inflammasome inhibitors and PPAR agonists—consistently reduces hepatic inflammation scores and simultaneously improves ballooning, steatosis and fibrosis, with some studies demonstrating histologic resolution of steatohepatitis. In clinical trials for NASH (e.g. obeticholic acid, selonsertib, lanifibranor), reduction in hepatic inflammation is a key primary or secondary endpoint and correlates with overall histologic improvement, further supporting the essentiality of hepatic inflammation for the AO in humans. Empirical support is strong.
- Quantitative understanding: Clinical and preclinical studies provide semi‑quantitative data showing that higher baseline inflammation scores (NAS inflammation sub‑score, lobular inflammation grade) are associated with greater likelihood and severity of steatohepatitis, and that reductions in inflammation score of ≥1 point correlate with overall histologic improvement. However, fully parameterised response–response functions that precisely translate a defined magnitude of hepatic inflammation into a predicted NAS or steatohepatitis severity score are not yet established, partly because of the composite and multi‑factorial nature of the AO definition. Quantitative understanding is therefore Moderate, being better supported than most intermediate KERs but not yet at the level required for formal qAOP modelling.
Known Modulating Factors
| Modulating Factor (MF) | Influence or Outcome | KER(s) involved |
|---|---|---|
| Diet composition (high fat/fructose, low fibre, choline deficiency) | Increases intestinal BA exposure and BA pool size, promotes hepatic steatosis and low‑grade inflammation; amplifies FGF15/19 suppression, CYP7A1 induction and severity of downstream inflammatory and barrier events, accelerating progression to the AO. | KER1, KER2, KER3, KER5, KER6, KER8, KER9, KER11 |
| Gut microbiota composition and function (dysbiosis) | Alters BA deconjugation/conversion (secondary BA formation), reduces SCFA production, impairs FXR/TGR5 signalling and mucus integrity; increases BA‑driven epithelial toxicity and PAMP leakage, enhancing hepatic inflammation via the gut–liver feedback loop. | KER5, KER6, KER7, KER8, KER10 |
| FXR–FGF15/19 axis genetic variation (NR1H4, FGF19, FGFR4, β‑Klotho) | Alters the sensitivity and gain of the FXR–FGF15/19–CYP7A1 feedback loop; determines how strongly a given level of intestinal FXR inhibition translates to FGF15/19 reduction, CYP7A1 derepression and intrahepatic BA burden. | KER1, KER2, KER3 |
| Systemic inflammation / circulating cytokines (TNF‑α, IL‑1β, IL‑6) | Represses BSEP expression and disrupts FXR signalling; impairs intestinal tight junctions and primes hepatic immune cells, amplifying both cholestatic and inflammatory progression along the AOP, including the liver‑to‑gut feedback. | KER4, KER5, KER10, KER11 |
| Concomitant liver insults (alcohol, hepatotoxic drugs, viral hepatitis) | Additively or synergistically increases intrahepatic BA retention, oxidative stress and hepatic inflammation; may lower the threshold at which upstream KEs lead to steatohepatitis. | KER3, KER4, KER9, KER11 |
Quantitative Understanding
Based on the evidence assembled across all KERs in this AOP, the overall quantitative understanding is semi‑quantitative and heterogeneous.
The proximal steps of the pathway are most consistently characterised in quantitative terms. Dose–response relationships between bile acid or FXR agonist/antagonist exposure and ileal FGF15/FGF19 expression and secretion have been demonstrated in vitro, in rodent ileal tissue and in human intervention studies (KER1). A robust inverse relationship between circulating FGF19 and serum C4 (a surrogate for hepatic CYP7A1 activity) has been documented in multiple human cohorts and pharmacologic studies, providing a reasonably well‑defined quantitative link spanning KER1 and KER2. Semi‑quantitative relationships between CYP7A1 activity and bile acid pool size (approximately two‑fold change in CYP7A1 yielding approximately two‑ to 2.5‑fold change in pool) are available from transgenic and knockout rodent models, supporting a moderate level of quantitative understanding for KER3.
For the intermediate KERs involving BSEP and bile acid composition (KER4, KER5), quantitative data are more limited. While mass spectrometry–based bile acid profiling allows precise compositional measurement, formal quantitative models linking defined changes in CYP7A1 activity or BA burden to specific shifts in species composition are not yet established. For KER6, concentration–response data for specific bile acids and intestinal barrier endpoints exist in vitro, but comprehensive in vivo quantitative functions are lacking. For KER7, correlations between permeability indices and portal endotoxin levels have been reported, but predictive quantitative models are not available.
For the more distal KERs (KER8 through KER11 and the feedback KER10), evidence is predominantly correlative or categorical. The magnitudes of oxidative stress and hepatic inflammation scale with the degree of BA overload and PAMP exposure, and intervention studies provide directional evidence, but precise dose–response and time–course functions applicable across species and metabolic contexts are sparse. The gut–liver feedback KER (KER10) has the weakest quantitative support, with only cross‑sectional clinical associations and no parameterised response–response function currently available.
Modulating factors such as diet, metabolic status, gut microbiota composition, sex and age introduce additional variability that complicates quantitative extrapolation, particularly to humans. Species differences in bile acid pool composition and FXR ligand potency further limit direct translation of rodent dose–response data.
Overall, while the direction and relative sensitivity of most KERs are supported by multiple datasets, fully parameterised response–response functions suitable for formal quantitative AOP (qAOP) modelling exist only for selected proximal parts of the pathway (KER1–KER3). The AOP is therefore currently best applied in a qualitative or semi‑quantitative manner to support mechanistic interpretation, hazard prioritisation and testing strategy design, rather than for precise numerical prediction of steatohepatitis incidence or severity.
Considerations for Potential Applications of the AOP (optional)
References
1. Simbrunner B, Hofer BS, Schwabl P, Zinober K, Petrenko O, Fuchs C, Semmler G, Marculescu R, Mandorfer M, Datz C, Trauner M, Reiberger T. FXR-FGF19 signaling in the gut-liver axis is dysregulated in patients with cirrhosis and correlates with impaired intestinal defence. Hepatol Int. 2024 Jun;18(3):929-942. doi: 10.1007/s12072-023-10636-4. Epub 2024 Feb 8. PMID: 38332428; PMCID: PMC11126514.
2. Hernandez GV, Smith VA, Melnyk M, Burd MA, Sprayberry KA, Edwards MS, Peterson DG, Bennet DC, Fanter RK, Columbus DA, Steibel JP, Glanz H, Immoos C, Rice MS, Santiago-Rodriguez TM, Blank J, VanderKelen JJ, Kitts CL, Piccolo BD, La Frano MR, Burrin DG, Maj M, Manjarin R. Dysregulated FXR-FGF19 signaling and choline metabolism are associated with gut dysbiosis and hyperplasia in a novel pig model of pediatric NASH. Am J Physiol Gastrointest Liver Physiol. 2020 Mar 1;318(3):G582-G609. doi: 10.1152/ajpgi.00344.2019. Epub 2020 Jan 31. PMID: 32003601; PMCID: PMC7099491.
3. Kliewer SA, Mangelsdorf DJ. Bile Acids as Hormones: The FXR-FGF15/19 Pathway. Dig Dis. 2015;33(3):327-31. doi: 10.1159/000371670. Epub 2015 May 27. PMID: 26045265; PMCID: PMC4465534.
4. Schumacher JD, Guo GL. Pharmacologic Modulation of Bile Acid-FXR-FGF15/FGF19 Pathway for the Treatment of Nonalcoholic Steatohepatitis. Handb Exp Pharmacol. 2019;256:325-357. doi: 10.1007/164_2019_228. PMID: 31201553; PMCID: PMC7033713.
5. Wen YQ, Zou ZY, Zhao GG, Zhang MJ, Zhang YX, Wang GH, Shi JJ, Wang YY, Song YY, Wang HX, Chen RY, Zheng DX, Duan XQ, Liu YM, Gonzalez FJ, Fan JG, Xie C. FXR activation remodels hepatic and intestinal transcriptional landscapes in metabolic dysfunction-associated steatohepatitis. Acta Pharmacol Sin. 2024 Nov;45(11):2313-2327. doi: 10.1038/s41401-024-01329-1. Epub 2024 Jul 11. PMID: 38992119; PMCID: PMC11489735.
6. Zhu S, Zou M, Wu Q, Zou Y, Tan T, Huang Z, Gong Z, Luo H, Dong X. The Gut-Liver Axis in Metabolic Dysfunction-Associated Steatotic Liver Disease: From Mechanistic Insights to Precision Therapeutics. FASEB J. 2026 Mar 31;40(6):e71687. doi: 10.1096/fj.202503607RR. PMID: 41824007; PMCID: PMC12986715.
7. Federico A, Dallio M, Godos J, Loguercio C, Salomone F. Targeting gut-liver axis for the treatment of nonalcoholic steatohepatitis: translational and clinical evidence. Transl Res. 2016 Jan;167(1):116-24. doi: 10.1016/j.trsl.2015.08.002. Epub 2015 Aug 12. PMID: 26318867.
8. Gil-Gómez A, Brescia P, Rescigno M, Romero-Gómez M. Gut-Liver Axis in Nonalcoholic Fatty Liver Disease: the Impact of the Metagenome, End Products, and the Epithelial and Vascular Barriers. Semin Liver Dis. 2021 May;41(2):191-205. doi: 10.1055/s-0041-1723752. Epub 2021 Mar 8. PMID: 34107545.
9. Cui C, Gao S, Shi J, Wang K. Gut-Liver Axis: The Role of Intestinal Microbiota and Their Metabolites in the Progression of Metabolic Dysfunction-Associated Steatotic Liver Disease. Gut Liver. 2025 Jul 15;19(4):479-507. doi: 10.5009/gnl240539. Epub 2025 May 8. PMID: 40336226; PMCID: PMC12261135.
10. Buzzetti E, Pinzani M, Tsochatzis EA. The multiple-hit pathogenesis of non-alcoholic fatty liver disease (NAFLD). Metabolism. 2016 Aug;65(8):1038-48. doi: 10.1016/j.metabol.2015.12.012. Epub 2016 Jan 4. PMID: 26823198.