
This AOP is licensed under the BY-SA license. This license allows reusers to distribute, remix, adapt, and build upon the material in any medium or format, so long as attribution is given to the creator. The license allows for commercial use. If you remix, adapt, or build upon the material, you must license the modified material under identical terms.
AOP: 575
Title
Decreased testosterone synthesis leading to increased nipple retention (NR) in male (rodent) offspring
Short name
Graphical Representation
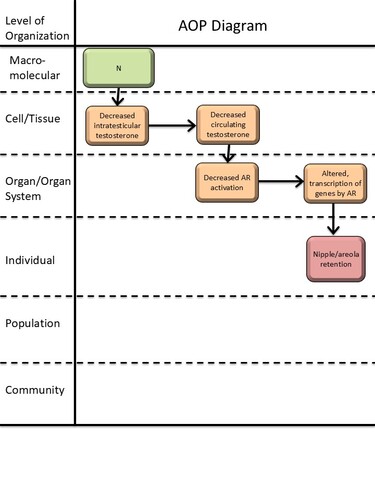
Point of Contact
Contributors
- Emilie Elmelund
- Terje Svingen
Coaches
OECD Information Table
| OECD Project # | OECD Status | Reviewer's Reports | Journal-format Article | OECD iLibrary Published Version |
|---|---|---|---|---|
| Scientific Review |
This AOP was last modified on March 20, 2026 10:50
Revision dates for related pages
| Page | Revision Date/Time |
|---|---|
| Decrease, intratesticular testosterone levels | March 20, 2026 12:04 |
| Decrease, circulating testosterone levels | March 20, 2026 12:12 |
| Decrease, androgen receptor activation | March 20, 2026 11:54 |
| Altered, Transcription of genes by the androgen receptor | March 20, 2026 12:02 |
| Nipple retention (NR), increased | March 21, 2026 09:18 |
| Decrease, intratesticular testosterone leads to nipple retention, increased | March 20, 2026 11:15 |
| Decrease, circulating testosterone levels leads to nipple retention, increased | March 20, 2026 11:20 |
| Decrease, intratesticular testosterone leads to Decrease, circulating testosterone levels | March 20, 2026 12:59 |
| Decrease, AR activation leads to nipple retention, increased | March 20, 2026 11:08 |
| Decrease, circulating testosterone levels leads to Decrease, AR activation | March 20, 2026 13:02 |
| Decrease, AR activation leads to Altered, Transcription of genes by the AR | March 20, 2026 10:30 |
| Dibutyl phthalate | November 29, 2016 18:42 |
| Di(2-ethylhexyl) phthalate | November 29, 2016 18:42 |
Abstract
This AOP links decreased intratesticular testosterone levels during fetal development with nipple/areola retention (NR) in male rodent offspring. NR, measured around 2 weeks postpartum, is a marker for disrupted masculinization of male offspring, with data primarily from laboratory mice and rats.
Testosterone is one of the two main steroid sex hormones essential for male reproductive development. Testosterone is primarily, but not exclusively, produced in the testes and then secreted into the circulation. In peripheral reproductive tissues, testosterone is either converted to dihydrotestosterone (DHT) or directly activates the androgen receptor (AR). AR is a nuclear receptor involved in the transcriptional regulation of various target genes during development and adulthood across species. AR signalling is necessary for normal masculinization of the developing fetus, and AR action in male rodents signals the nipple anlagen to regress, leaving males with no nipples.
This AOP delineates the evidence that decreasing testicular testosterone production lowers circulating testosterone levels and consequently AR activation, thereby causing retention of nipples in male rodents. In this AOP, the first KE is not considered an MIE, as testicular testosterone production can be obstructed by various mechanisms (Miller & Auchus, 2011). Moreover, the AOP does not discriminate whether the reduction in AR activation is due to a direct lack of testosterone binding AR or due to decreased conversion of testosterone to DHT, as there is not sufficient information on this distinction. Downstream of a reduction in AR activation, the molecular mechanisms of NR are unclear, highlighting a knowledge gap in this AOP and potential for further development.
The confidence in KER-3486 (‘Decrease, circulating testosterone levels’ leads to ‘Increase, nipple retention’) is moderate due to the limited empirical evidence available. The confidence in each of the remaining KERs comprising the AOP is judged as high, with both high biological plausibility and high confidence in empirical evidence. The mechanistic link between KE-286 (‘altered, transcription of genes by AR’) and AO-1786 (‘increase, nipple retention’) is not established, but given the high confidence in the KERs, the overall confidence in the AOP is judged as high.
The AOP supports the regulatory application of NR as a measure of endocrine disruption relevant for human health and the use of NR as an indicator of anti-androgenicity in environmentally relevant species. Even though NR cannot be directly translated to a human endpoint, the AOP is considered human relevant since NR is a clear readout of reduced androgen action and fetal masculinization during development and is considered an ‘adverse outcome’ in OECD test guidelines (TG 443, TG 421, TG 422). The AOP also holds utility for informing on anti-androgenicity more generally, as this modality is highly relevant across mammalian species.
AOP Development Strategy
Context
This AOP is a part of an AOP network for reduced AR activation leading to increased NR in male offspring. The other AOPs in this network are AOP-344 (‘Androgen receptor antagonism leading to increased nipple retention (NR) in male (rodent) offspring’) and AOP 576 (‘5α-reductase inhibition leading to increased nipple retention (NR) in male (rodent) offspring’). The purpose of the AOP network is to organize the well-established evidence for anti-androgenic mechanisms-of-action leading to increased NR. It can be used in the identification and assessment of endocrine disruptors and to inform predictive toxicology, identification of knowledge gaps for investigation and method development.
This work received funding from the European Food and Safety Authority (EFSA) under Grant agreement no. GP/EFSA/PREV/2022/01.
Strategy
The OECD AOP Developer’s Handbook v2.7was followed alongside pragmatic approaches (Svingen et al., 2021).
The adverse outcome, AO 1786: Increase nipple retention, was originally developed in (Pedersen et al., 2022).
Some upstream KEs and KERs were developed in Draskau et al., 2024 as part of an anti-androgenic network, using mainly key review publications, since it was considered canonical knowledge. This included KE 1690: Decrease, circulating testosterone levels; KE 1614: Decrease, AR activation and KE 286: Altered transcription of genes by the AR as well as the connecting KERs, KER 2131: Decrease, circulating testosterone levels leads to decrease, AR activation and KER 2124: Decrease, AR activation leads to altered transcription of genes by AR (Draskau et al., 2024).
KE 2298 (Decrease, intratesticular testosterone levels) and KER 3448, connecting this to KE 1690 (Decrease, intratesticular testosterone levels leads to decrease, circulation testosterone levels) were added as part of an AOP network, ultimately leading to the AO decreased anogenital distance (Svingen et al., 2025). This was done to discriminate between the large difference in testosterone levels between the testes and in circulation (Coviello et al., 2004). As for other upstream KEs and KERs, this was considered canonical knowledge, and the KE and KER were developed using mainly key review publications (Draskau et al., 2024).
The non-adjacent KERs 3487, 3486, and 3348 linking reduced decreased intratesticular testosterone, circulating testosterone, and AR activation with increased NR, respectively, were developed using a systematic weight-of-evidence approach, following the methodology outlined in (Holmer et al., 2024). Publications were retrieved by literature searches in PubMed and Web of Science and extensive screening using pre-defined inclusion and exclusion criteria. Evaluation of methodological reliability of in vivo animal studies was performed using the Science in Risk Assessment and Policy (SciRAP) online tool. For KERs 3487 and 3486 regarding testosterone levels, publications were included if there was a decrease in fetal testosterone levels and NR was assessed in male offspring. For KER 3348, there are currently no in vivo methods to measure AR activation in mammals, and instead, six chemicals with known anti-androgenic mechanisms-of-action were chosen for the empirical evidence for this KER.
The rationale for the inclusion of KEs and KERs in the upstream anti-androgenic network is detailed in (Draskau et al., 2024). The link between the upstream network, more specifically KE-286 (‘altered, transcription of genes by AR’), and AO-1786 (‘increase, nipple retention’) likely contains a tissue-specific KE that has not been developed, as sufficient evidence is not yet available. Thus, for now, the most evidence for the link between the upstream anti-androgenic network and increased nipple retention is captured by KERs 3348, 3486 and 3487.
Summary of the AOP
Events:
Molecular Initiating Events (MIE)
Key Events (KE)
Adverse Outcomes (AO)
| Type | Event ID | Title | Short name |
|---|
| KE | 2298 | Decrease, intratesticular testosterone levels | Decrease, intratesticular testosterone |
| KE | 1690 | Decrease, circulating testosterone levels | Decrease, circulating testosterone levels |
| KE | 1614 | Decrease, androgen receptor activation | Decrease, AR activation |
| KE | 286 | Altered, Transcription of genes by the androgen receptor | Altered, Transcription of genes by the AR |
| AO | 1786 | Nipple retention (NR), increased | nipple retention, increased |
Relationships Between Two Key Events (Including MIEs and AOs)
| Title | Adjacency | Evidence | Quantitative Understanding |
|---|
| Decrease, intratesticular testosterone leads to nipple retention, increased | non-adjacent | High | |
| Decrease, circulating testosterone levels leads to nipple retention, increased | non-adjacent | Moderate | |
| Decrease, AR activation leads to nipple retention, increased | non-adjacent | High |
Network View
Prototypical Stressors
Life Stage Applicability
| Life stage | Evidence |
|---|---|
| Foetal | High |
Taxonomic Applicability
Sex Applicability
| Sex | Evidence |
|---|---|
| Male | High |
Overall Assessment of the AOP
Domain of Applicability
The upstream part of the AOP has a broad applicability domain, but the downstream KERs 3487 (‘Decrease, intratesticular testosterone levels leads to increase, nipple retention’), 3486 (‘Decrease, circulating testosterone levels leads to increase, nipple retention’), and 3348 (‘Decrease AR activation leads to increase, nipple retention’) are considered only directly applicable to male rodents (current evidence stems primarily from laboratory rats and mice) during fetal life, restricting the taxonomic applicability of the AOP. Although NR has primarily been investigated in rats and mice, it is biologically plausible that the AOP is applicable to other rodent species. The process of retention of nipples by disruption of androgen programming happens in the fetal life stage, but the AO is detected postnatally. Specifically, the MPW (~gestational days (GD) 16-20 in rat, presumably gestational weeks (GW) 8-14 in humans) is the primary fetal window of applicability, but effects outside of this window in fetal life, after androgen production has started, cannot be excluded. In the males of these species, the nipple anlagen are programmed during fetal development by androgens to regress, leading to no visible nipples in males postnatally, while female rats and mice exhibit nipples. This AOP only contains empirical evidence for the applicability to male rats, but the AOP is considered equally applicable to male mice as these also normally exhibit nipple regression stimulated by androgens. Moreover, the AOP is relevant for other taxa, including humans, as NR in male rodents indicates a reduction in fetal masculinization. NR is therefore included as a mandatory endpoint in multiple OECD Test Guideline studies for developmental and reproductive toxicity and is considered applicable as an adverse outcome to set NOAELs and LOAELs of substances in human health risk assessments.
Essentiality of the Key Events
|
Event |
Evidence |
Uncertainties and inconsistencies |
|
KE-2298 Decreased, intratesticular testosterone (ITT) levels MODERATE: Testis is the primary organ in males for testosterone synthesis and is required for serum testosterone. Studies with exposure to phthalates show reduced ITT levels and increased nipple retention.
|
Biological plausibility provides strong support for the essentiality of this event, as the testes are the primary testosterone producing organs in male mammals and testosterone is a ligand of the AR and a main driver for normal regression of nipple anlagen in male offspring (Goldman et al., 1976). Indirect evidence of impact of decreased ITT (KE-2298) on decreased circulating T (KE-1690) • Castrated males have significantly reduced serum T. Although at different life stage, it is highly likely same relationship exists in fetal males, with loss of testosterone from testis resulting in loss of circulating testosterone. Indirect evidence of impact of decreased ITT (KE-2298) on increased nipple retention (AO-1786): • Numerous rat studies evidence a relationship between reduced intratesticular testosterone levels caused by exposure to phthalates and increased nipple retention in male offspring (see empirical evidence table in KER-3487). |
|
|
KE-1690 Decreased, circulating testosterone (CT) levels MODERATE: CT is substrate for DHT production, also locally, and numerous studies have shown strong relationships between reduced CT and increased nipple retention. |
Biological plausibility provides strong support for the essentiality of this event. Testosterone is an AR ligand and a main driver for regression of nipple anlagen in male offspring (Goldman et al., 1976), as well as a substrate for local production of DHT (Imperato-McGinley J et al., 1986). Indirect evidence of the impact of decreased CT (KE-1690) on AR activity in vitro: • Increasing concentrations of testosterone lead to increasing AR activation in vitro (U. S. EPA, 2018) (see also KER-2131). Indirect evidence of impact of decreased CT (KE-1690) on increased nipple retention (AO-1786): • Exposure to the phthalates DEHP and DBP during prenatal development in rats results in reduced fetal testosterone levels and increased nipple retention in male offspring. Literature review on the relationship has judged the link to be strongly evidenced (See empirical evidence in KER-3348). Indirect evidence of the impact of decreased CT (KE-1690) on increased nipple retention (AO-1786): • Nipple formation is inhibited in female rat fetuses exposed to testosterone during gestation (Goldman et al., 1976). |
Inconsistencies in indirect evidence of impact on the AO: • Some inconsistencies were observed in the empirical evidence regarding increased nipple retention in male pups after in utero exposure to DEHP. However, all inconsistencies could be explained by differences in exposure doses and statistical power (See empirical evidence in KER-3348)
|
|
KE-1614 Decreased, AR activation HIGH: There is experimental evidence from mutant mice insensitive to androgens showing that the AR is essential for nipple retention in male offspring. There is also evidence from exposure studies in animals that substances antagonizing AR induce nipple retention in male pups. |
Biological plausibility provides strong support for the essentiality of this event, as AR activation is critical for normal regression of nipple anlagen in male embryos. Indirect evidence of the impact of decreased AR activation (KE-1614) on altered gene transcription by AR (KE-286): • Exposure to known anti-androgenic chemicals induces a changed gene expression pattern, e.g. in neonatal pig ovaries (Knapczyk-Stwora et al., 2019). Direct evidence of the impact of decreased AR activation (KE-1614) on altered gene transcription by AR (KE-286): • Male AR KO mice have altered gene expression pattern in a broad range of organs (refer to KER-2124). Indirect evidence of impact of decreased AR activation (KE-1614) on increased nipple retention (AO-1786): • Rat in vivo exposure to vinclozolin, procymidone and flutamide, which are known AR antagonists, leads to increased nipple retention in offspring (see KER-3348). Direct evidence of impact of decreased AR activation (KE-1614) on increased nipple retention (AO-1786): • Male Tfm mutant mice, which are insensitive to androgens and believed to be so due to a nonfunctional androgen receptor, present with retained nipples (Kratochwil & Schwartz, 1976) |
|
|
KE-286 Altered, trans. of genes by AR LOW: Strongest support for essentiality comes from biological plausibility. However, exact transcriptional effects and causality remain to be fully characterized. |
Biological plausibility provides support for the essentiality of this event. AR is a nuclear receptor and transcription factor regulating transcription of genes, and androgens, acting through AR, are essential for normal regression of nipple anlagen in male fetuses. |
There are currently no AR-responsive genes proven to be causally involved in nipple retention, and it is known that AR can also signal through non-genomic actions (Leung & Sadar, 2017). |
|
Event |
Direct evidence |
Indirect evidence |
Contradictory evidence |
Overall essentiality assessment |
|
KE-2298 |
|
*** |
|
Moderate |
|
KE-1690 |
|
*** |
|
Moderate |
|
KE-1614 |
*** |
*** |
|
High |
|
KE-286 |
|
|
|
Low (biological plausibility) |
*Low level of evidence (some support for essentiality), ** Intermediate level of evidence (evidence for impact on one or more downstream KEs), ***High level of evidence (evidence for impact on AO).
Evidence Assessment
Confidence in KER-3486 is considered moderate due to the limited empirical evidence available. The confidence in each of the remaining KERs comprising the AOP is judged as high, with both high biological plausibility and high confidence in empirical evidence. The mechanistic link between KE-286 (‘altered, transcription of genes by AR’) and AO-1786 (‘increase, nipple retention’) is not established, but given the high confidence in the KERs, the overall confidence in the AOP is judged as high.
|
KER |
Biological Plausibility |
Empirical Evidence |
Rationale |
|
KER-3448 Decrease, intratesticular testosterone levels leads to a decrease, circulating testosterone levels |
High |
High (canonical) |
It is well established that testes are the primary testosterone-producing organs in male mammals. In vivo studies have shown that exposure to substances that lower intratesticular testosterone also lowers circulating testosterone levels (Svingen et al., 2025). |
|
KER-2131 Decrease, circulating testosterone levels leads to decrease, AR activation |
High |
High (canonical) |
It is well established that testosterone activates the AR. Direct evidence for this KER is not possible since KE 1614 can currently not be measured and is considered an in vivo effect. Indirect evidence using proxy read-outs of AR activation, either in vitro or in vivo strongly supports the relationship (Draskau et al., 2024) |
|
KER-2124 Decrease, AR activation leads to altered, transcription of genes by AR |
High |
High (canonical) |
It is well established that the AR regulates gene transcription. In vivo animal studies and human genomic profiling show tissue-specific changes to gene expression upon disruption of AR (Draskau et al., 2024). |
|
KER-3487 Decrease, intratesticular testosterone leads to an increase, nipple retention |
High |
High |
It is well established that testicular testosterone is one of the primary androgens responsible for the regression of nipple anlagen in male rodent fetuses In vivo animal studies support that reductions in fetal testicular testosterone can cause NR in male offspring. Temporal concordance is generally supported, while dose concordance is more weakly suggested. |
|
KER-3486 Decrease, circulating testosterone levels leads to increase, nipple retention |
High |
Moderate |
It is well established that testosterone is one of the primary androgens responsible for the regression of nipple anlagen in male rodent fetuses Two in vivo rat toxicity studies support the relationship and temporal concordance of the KER. Dose concordance is not informed. |
|
KER-3348 Decrease, AR activation leads to increase, nipple retention |
High |
High |
It is well established that activation of AR regression of nipple anlagen in males. The empirical evidence includes numerous in vivo toxicity studies showing that decreased AR activation leads to increased NR in male offspring, with few inconsistencies. Empirical evidence combined with theoretical considerations provide some support for dose, temporal, and incidence concordance for the KER, although this evidence is weak and indirect. It should be recognized that the upstream KE-1614 cannot currently be measured directly (in vivo). Instead, empirical evidence was therefore collected for substances known to affect upstream events. This limitation is not considered to lower the strength of the evidence in this case. |
Known Modulating Factors
|
Modulating factor (MF) |
Influence or Outcome |
KER(s) involved |
|
Genotype |
Extended CAG repeat length in AR is associated with reduced AR activity (Chamberlain et al., 1994; Tut et al., 1997). This MF could initiate the AOP at lower stressor doses. |
KER 2131, KER 2124, KER 3348 |
|
Rat strain |
Long-Evans Hooded rat is less sensitive to NR than the Sprague-Dawley rat (Wolf et al., 1999; You L et al., 1998). Thus, the effects on AO at certain stressor doses may vary between strains |
KER 3487, KER 3486, KER 3348
|
| Adrenal species difference | Adrenal glands can supply weak androgen precursors, contributing to circulating androgen levels, although not substituting for testicular testosterone in masculinization processes. The magnitude and mechanism of adrenal androgen synthesis is also species dependent (Olson & Ristau, 2025; Pihlajoki et al., 2015). | KER-3448 |
Quantitative Understanding
The quantitative understanding of this AOP is judged as low.
A model for phthalate-induced malformations has been developed which aims to predict the degree of NR related to a phthalate’s reduction in ex vivo testosterone production. The model predicted that a 40% reduction in testosterone levels would induce NR in male rats, with increasing number of nipples as testosterone levels decrease (Gray et al., 2024).
Considerations for Potential Applications of the AOP (optional)
The AOP supports the regulatory application of NR as a measure of endocrine disruption relevant for human health and the use of NR as an indicator of anti-androgenicity in environmentally relevant species. NR is a mandatory endpoint in multiple OECD test guidelines, including TG 443 (extended one-generation reproductive toxicity study) and TGs 421/422 (reproductive toxicity screening studies) (OECD 2025a; OECD 2025b; OECD 2025c). NR can contribute to establishing a No Observed Adverse Effect Level (NOAEL), as outlined in OECD guidance documents No. 43 and 151 (OECD 2008; OECD 2013). The ability to derive a NOAEL for increased NR in male rodent offspring, which can serve as a point of departure for determining human safety thresholds, underscores the regulatory significance of this AOP. The AOP also holds utility for informing on anti-androgenicity more generally, as this modality is highly relevant across mammalian species (Schwartz et al., 2021).
References
Chamberlain, N. L., Driver, E. D., & Miesfeld, R. L. (1994). The length and location of CAG trinucleotide repeats in the androgen receptor N-terminal domain affect transactivation function. Nucleic Acids Research, 22(15), 3181–3186. https://doi.org/10.1093/nar/22.15.3181
Coviello, A. D., Bremner, W. J., Matsumoto, A. M., Herbst, K. L., Amory, J. K., Anawalt, B. D., Yan, X., Brown, T. R., Wright, W. W., Zirkin, B. R., & Jarow, J. P. (2004). Intratesticular Testosterone Concentrations Comparable With Serum Levels Are Not Sufficient to Maintain Normal Sperm Production in Men Receiving a Hormonal Contraceptive Regimen. Journal of Andrology, 25(6), 931–938. https://doi.org/10.1002/j.1939-4640.2004.tb03164.x
Draskau, M. K., Rosenmai, A. K., Bouftas, N., Johansson, H. K. L., Panagiotou, E. M., Holmer, M. L., Elmelund, E., Zilliacus, J., Beronius, A., Damdimopoulou, P., van Duursen, M., & Svingen, T. (2024). AOP Report: An Upstream Network for Reduced Androgen Signaling Leading to Altered Gene Expression of Androgen Receptor–Responsive Genes in Target Tissues. Environmental Toxicology and Chemistry, 43(11), 2329–2337. https://doi.org/10.1002/etc.5972
Gray, L. E. J., Lambright, C. S., Evans, N., Ford, J., & Conley, J. M. (2024). Using targeted fetal rat testis genomic and endocrine alterations to predict the effects of a phthalate mixture on the male reproductive tract. Current Research in Toxicology, 7, 100180. https://doi.org/10.1016/j.crtox.2024.100180
Goldman AS, Shapiro B, & Neumann F. (1976). Role of testosterone and its metabolites in the differentiation of the mammary gland in rats. Endocrinology, 99(6), 1490–1495. https://doi.org/10.1210/endo-99-6-1490
Holmer, M. L., Zilliacus, J., Draskau, M. K., Hlisníková, H., Beronius, A., & Svingen, T. (2024). Methodology for developing data-rich Key Event Relationships for Adverse Outcome Pathways exemplified by linking decreased androgen receptor activity with decreased anogenital distance. Reproductive Toxicology, 128, 108662. https://doi.org/10.1016/j.reprotox.2024.108662
Imperato-McGinley J, Binienda Z, Gedney J, & Vaughan ED Jr. (1986). Nipple differentiation in fetal male rats treated with an inhibitor of the enzyme 5 alpha-reductase: definition of a selective role for dihydrotestosterone. Endocrinology, 118(1), 132–137. https://doi.org/10.1210/endo-118-1-132
Knapczyk-Stwora, K., Nynca, A., Ciereszko, R. E., Paukszto, L., Jastrzebski, J. P., Czaja, E., Witek, P., Koziorowski, M., & Slomczynska, M. (2019). Flutamide-induced alterations in transcriptional profiling of neonatal porcine ovaries. Journal of Animal Science and Biotechnology, 10(1), 35. https://doi.org/10.1186/s40104-019-0340-y
Kratochwil, K., & Schwartz, P. (1976). Tissue interaction in androgen response of embryonic mammary rudiment of mouse: identification of target tissue for testosterone. Proceedings of the National Academy of Sciences, 73(11), 4041–4044. https://doi.org/10.1073/pnas.73.11.4041
Leung, J. K., & Sadar, M. D. (2017). Non-Genomic Actions of the Androgen Receptor in Prostate Cancer. Frontiers in Endocrinology, 8. https://doi.org/10.3389/fendo.2017.00002
Miller, W. L., & Auchus, R. J. (2011). The Molecular Biology, Biochemistry, and Physiology of Human Steroidogenesis and Its Disorders. Endocrine Reviews, 32(1), 81–151. https://doi.org/10.1210/er.2010-0013
OECD (2008), Guidance Document on Mammalian Reproductive Toxicity Testing and Assessment, OECD Series on Testing and Assessment, No. 43, OECD Publishing, Paris, https://doi.org/10.1787/d2631d22-en.
OECD (2013), Guidance Document Supporting OECD Test Guideline 443 on the Extended One-Generational Reproductive Toxicity Test, OECD Series on Testing and Assessment, No. 151, OECD Publishing, Paris, ENV/JM/MONO(2013)10
OECD. (2025a). Test No. 421: Reproduction/Developmental Toxicity Screening Test. https://doi.org/10.1787/9789264264380-en
OECD. (2025b). Test No. 422: Combined Repeated Dose Toxicity Study with the Reproduction/Developmental Toxicity Screening Test. https://doi.org/10.1787/9789264264403-en
OECD. (2025c). Test No. 443: Extended One-Generation Reproductive Toxicity Study. https://doi.org/10.1787/9789264185371-en
Olson, P., & Ristau, B. T. (2025). Review of Adrenal Androgen Synthesis, Hypersecretion, and Blockade. Urologic Clinics of North America, 52(2), 217–227. https://doi.org/10.1016/j.ucl.2025.01.004
Pihlajoki, M., Dörner, J., Cochran, R. S., Heikinheimo, M., & Wilson, D. B. (2015). Adrenocortical Zonation, Renewal, and Remodeling. Frontiers in Endocrinology, 6. https://doi.org/10.3389/fendo.2015.00027
Pedersen, E. B., Christiansen, S., & Svingen, T. (2022). AOP key event relationship report: Linking androgen receptor antagonism with nipple retention. In Current Research in Toxicology (Vol. 3). Elsevier B.V. https://doi.org/10.1016/j.crtox.2022.100085
Schwartz, C. L., Christiansen, S., Hass, U., Ramhøj, L., Axelstad, M., Löbl, N. M., & Svingen, T. (2021). On the Use and Interpretation of Areola/Nipple Retention as a Biomarker for Anti-androgenic Effects in Rat Toxicity Studies. In Frontiers in Toxicology (Vol. 3). Frontiers Media S.A. https://doi.org/10.3389/ftox.2021.730752
Svingen, T., Villeneuve, D. L., Knapen, D., Panagiotou, E. M., Draskau, M. K., Damdimopoulou, P., & O’Brien, J. M. (2021). A Pragmatic Approach to Adverse Outcome Pathway Development and Evaluation. Toxicological Sciences, 184(2), 183–190. https://doi.org/10.1093/toxsci/kfab113
Svingen T, Elmelund E, Holmer ML, Bindel AO, Holbech H, Draskau MK. AOP report: Adverse Outcome Pathway Network for Developmental Androgen Signalling-Inhibition Leading to Short Anogenital Distance in Male Offspring. Environ Toxicol Chem. 2025 Sep 1:vgaf221. doi: 10.1093/etojnl/vgaf221. Epub ahead of print. PMID: 40888748.
Tut, T. G., Ghadessy, F. J., Trifiro, M. A., Pinsky, L., & Yong, E. L. (1997). Long Polyglutamine Tracts in the Androgen Receptor Are Associated with Reduced Trans -Activation, Impaired Sperm Production, and Male Infertility 1. The Journal of Clinical Endocrinology & Metabolism, 82(11), 3777–3782. https://doi.org/10.1210/jcem.82.11.4385
U. S. EPA. (2018, October). ToxCast & Tox21 AR agonism of testosterone. https://www.epa.gov/comptox-tools/exploring-toxcast-data
Wolf, C., Lambright, C., Mann, P., Price, M., Cooper, R. L., Ostby, J., & Earl Gray, L. J. (1999). Administration of potentially antiandrogenic pesticides (procymidone, linuron, iprodione, chlozolinate, p,p-DDE, and ketoconazole) and toxic substances (dibutyl-and diethylhexyl phthalate, PCB 169, and ethane dimethane sulphonate) during sexual differentiation produces diverse profiles of reproductive malformations in the male rat. Toxicology and Industrial Health, 15, 94–118. www.stockton-press.co.uk
You L, Casanova M, Archibeque-Engle S, Sar M, Fan LQ, & Heck HA. (1998). Impaired male sexual development in perinatal Sprague-Dawley and Long-Evans hooded rats exposed in utero and lactationally to p,p’-DDE. Toxicological Sciences : An Official Journal of the Society of Toxicology, 45(2), 162–173. https://doi.org/10.1093/toxsci/45.2.162