AOP ID and Title:
Graphical Representation
Status
| Author status | OECD status | OECD project | SAAOP status |
|---|---|---|---|
| Under development: Not open for comment. Do not cite |
Abstract
This AOP links in utero androgen receptor (AR) antagonism with hypospadias in male offspring. Hypospadias is a common reproductive disorder with a prevalence of up to ~1/125 newborn boys (Leunbach et al., 2025; Paulozzi, 1999). Developmental exposure to endocrine disrupting chemicals is suspected to contribute to some cases of hypospadias (Mattiske & Pask, 2021). Hypospadias can be indicative of fetal disruptions to male reproductive development, and is associated with short anogenital distance and cryptorchidism (Skakkebaek et al., 2016). Thus, hypospadias is included as an endpoint in OECD test guidelines (TG) for developmental and reproductive toxicity (TG 414, 416, 421/422, and 443; (OECD, 2016b, 2016a, 2018a, 2018b, 2021)), as both a measurement of adverse reproductive effects and a direct clinical adverse outcome. In normal male reproductive development, androgen activation of the AR plays an essential role in driving differentiation of the male phenotype, including development of the penis (Amato et al., 2022). This AOP delineates the evidence that antagonism of AR through a reduction in AR activation can disrupt penis development and cause hypospadias. Substantial evidence exists that links exposure to AR antagonists to hypospadias in in vivo rodent studies. Moreover, human case studies of subjects with AR mutations and hypospadias support the link. Downstream of a reduction in AR activation, the molecular mechanisms of hypospadias development are less clear, highlighting a knowledge gap in this AOP. Thus, the AOP has potential for inclusion of additional KEs and elaboration of molecular causality links, once these are established. Given that hypospadias is both a clinical and toxicological endpoint, this AOP is considered highly relevant in a regulatory context.
Background
This AOP is a part of an AOP network for reduced androgen receptor activation causing hypospadias in male offspring. The other AOPs in this network are AOP-570 (‘Decreased testosterone synthesis leading to hypospadias in male (mammalian) offspring’) and AOP-571 (‘5α-reductase inhibition leading to hypospadias in male (mammalian) offspring’). The purpose of the AOP network is to organize the well-established evidence for anti-androgenic mechanisms-of-action leading to hypospadias, thus informing predictive toxicology and identifying knowledge gaps for investigation and method development.
This work received funding from the European Food and Safety Authority (EFSA) under Grant agreement no. GP/EFSA/PREV/2022/01 and from the Danish Environmental Protection Agency under the Danish Center for Endocrine Disrupters (CeHoS).
Summary of the AOP
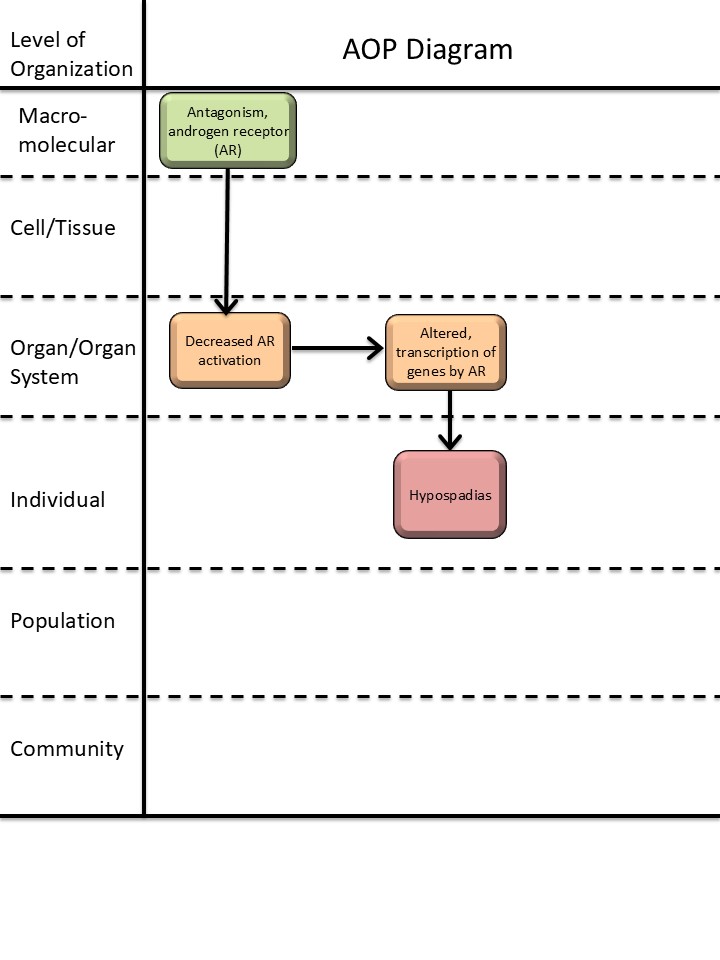
Events
Molecular Initiating Events (MIE), Key Events (KE), Adverse Outcomes (AO)
| Sequence | Type | Event ID | Title | Short name |
|---|---|---|---|---|
| MIE | 26 | Antagonism, Androgen receptor | Antagonism, Androgen receptor | |
| KE | 1614 | Decrease, androgen receptor activation | Decrease, AR activation | |
| KE | 286 | Altered, Transcription of genes by the androgen receptor | Altered, Transcription of genes by the AR | |
| AO | 2082 | Hypospadias, increased | Hypospadias |
Key Event Relationships
| Upstream Event | Relationship Type | Downstream Event | Evidence | Quantitative Understanding |
|---|---|---|---|---|
| Antagonism, Androgen receptor | adjacent | Decrease, androgen receptor activation | High | |
| Decrease, androgen receptor activation | adjacent | Altered, Transcription of genes by the androgen receptor | High | |
| Decrease, androgen receptor activation | non-adjacent | Hypospadias, increased | High |
Stressors
| Name | Evidence |
|---|---|
| Flutamide | |
| Vinclozolin | |
| Procymidone |
Overall Assessment of the AOP
Domain of Applicability
Life Stage Applicability| Life Stage | Evidence |
|---|---|
| Foetal | High |
| Term | Scientific Term | Evidence | Links |
|---|---|---|---|
| human | Homo sapiens | High | NCBI |
| rat | Rattus norvegicus | High | NCBI |
| mouse | Mus musculus | Moderate | NCBI |
| Sex | Evidence |
|---|---|
| Male | High |
Although the upstream part of the AOPN has a broad applicability domain, the overall AOPN is considered only applicable to male mammals during fetal life, restricted by the applicability of KER-2828 (‘Decrease, AR activation leads to hypospadias’). The term hypospadias is mainly used for describing malformation of the male, and not female, external genitalia. Some studies refer to hypospadias in females, but these have not been reported to be caused by exposure to AR antagonists, and the mechanisms behind these malformations are likely different from the mechanisms in males (Greene, 1937; Stewart et al., 2018). The genital tubercle is programmed by androgens to differentiate into a penis in fetal life during the masculinization programming window, followed by the morphological differentiation (Welsh et al., 2008). In humans, hypospadias is diagnosed at birth and can also often be observed in rodents (rats and mice) at this time point, although the rodent penis does not finish developing until a few weeks after birth (Baskin & Ebbers, 2006; Sinclair et al., 2017). The disruption to androgen programming leading to hypospadias thus takes place during fetal life, but the AO itself is best detected postnatally. Regarding taxonomic applicability, hypospadias has mainly been described in rodents and humans, and the evidence in this AOP is almost exclusively from these species. It is, however, biologically plausible that the AOP is applicable to other mammals, given the conserved role of androgens in mammalian reproductive development, and hypospadias has been observed in many domestic animal and wildlife species, albeit not coupled to AR antagonism.
Essentiality of the Key Events
|
Event |
Evidence |
Uncertainties and inconsistencies |
|
MIE-26 Antagonism, AR (high) |
Biological plausibility provides strong support for the essentiality of this event, as androgens, acting through AR, are the primary drivers of penis development In vivo toxicity studies in rats show that in utero exposure to known AR antagonists flutamide, procymidone, and vinclozolin cause hypospadias in male offspring (listed in table 3 in KER 2828) Ex vivo culture of genital tubercles with AR antagonist flutamide disrupted genital tubercle development, but this was rescued with addition of DHT (Petiot et al., 2005) |
In the ex vivo study, androgens were not added to the baseline culture media, even though they are present in an in vivo scenario to induce penis differentiation. |
|
KE-1614 Decrease, AR activation (moderate) |
Biological plausibility provides strong support for the essentiality of this event, as AR activation is critical for normal penis development.
Conditional or full knockout of Ar in mice results in partly or full sex-reversal of males, including a female-like urethral opening(Willingham et al., 2006; Yucel et al., 2004; Zheng et al., 2015). Human subjects with AR mutations may also have associated hypospadias (as listed in table 4 in KER 2828). |
|
|
KE-286 Altered, transcription of genes by AR (low) |
Biological plausibility provides support for the essentiality of this event. AR is a nuclear receptor and transcription factor regulating transcription of genes, and androgens, acting through AR, are essential for normal male penis development. Known AR-responsive genes active in normal penis development have been thoroughly reviewed (Amato et al., 2022). |
There are currently no AR-responsive genes proved to be causally involved in hypospadias, and it is known that the AR can also signal through non-genomic actions (Leung & Sadar, 2017). |
|
Event |
Direct evidence |
Indirect evidence |
Contradictory evidence |
Overall essentiality assessment |
|
MIE-26 |
*** |
|
|
High |
|
KE-1614 |
** |
|
|
Moderate |
|
KE-286 |
|
* |
|
Low |
Weight of Evidence Summary
The confidence in each of the KERs comprising the AOP are judged as high, with both high biological plausibility and high confidence in the empirical evidence. The mechanistic link between KE-286 (‘altered, transcription of genes by AR’) and AO-2082 (‘hypospadias’) is not established, but given the high confidence in the KERs including the non-adjacent KER-2828 linking to the AO, the overall confidence in the AOP is judged as high.
|
KER |
Biological Plausibility |
Empirical Evidence |
Rationale |
|
KER-2130 Antagonism, AR leads to decrease, AR activation |
High |
High (canonical) |
It is well established that antagonism of the AR leads to decreased AR activity, and this has been evidenced in vitro (Draskau et al., 2024; Pedersen et al., 2022). |
|
KER-2124 Decrease, AR activation leads to altered, transcription of genes by AR |
High |
High (canonical) |
It is well established that the AR regulates gene transcription. In vivo animal studies and human genomic profiling show tissue-specific changes to gene expression upon disruption of AR (Draskau et al., 2024). |
|
KER-2828 Decrease, AR activation leads to hypospadias |
High |
High |
It is well established that AR drives penis differentiation. Numerous in vivo toxicity studies and human case studies indirectly show that decreased AR activation leads to hypospadias, with few inconsistencies. The empirical evidence moderately supports dose, temporal, and incidence concordance for the KER. |
Quantitative Consideration
The quantitative understanding of this AOP is judged as low.
References
Amato, C. M., Yao, H. H.-C., & Zhao, F. (2022). One Tool for Many Jobs: Divergent and Conserved Actions of Androgen Signaling in Male Internal Reproductive Tract and External Genitalia. Frontiers in Endocrinology, 13, 910964. https://doi.org/10.3389/fendo.2022.910964
Baskin, L., & Ebbers, M. (2006). Hypospadias: Anatomy, etiology, and technique. Journal of Pediatric Surgery, 41(3), 463–472. https://doi.org/10.1016/j.jpedsurg.2005.11.059
Chamberlain, N. L., Driver, E. D., & Miesfeld, R. L. (1994). The length and location of CAG trinucleotide repeats in the androgen receptor N-terminal domain affect transactivation function. Nucleic Acids Research, 22(15), 3181–3186. https://doi.org/10.1093/nar/22.15.3181
Draskau, M., Rosenmai, A., Bouftas, N., Johansson, H., Panagiotou, E., Holmer, M., Elmelund, E., Zilliacus, J., Beronius, A., Damdimopoulou, P., van Duursen, M., & Svingen, T. (2024). Aop Report: An Upstream Network for Reduced Androgen Signalling Leading to Altered Gene Expression of Ar Responsive Genes in Target Tissues. Environ Toxicol Chem, In Press.
Greene, R. R. (1937). Production of Experimental Hypospadias in the Female Rat. Proceedings of the Society for Experimental Biology and Medicine, 36(4), 503–506. https://doi.org/10.3181/00379727-36-9287P
Holmer, M. L., Zilliacus, J., Draskau, M. K., Hlisníková, H., Beronius, A., & Svingen, T. (2024). Methodology for developing data-rich Key Event Relationships for Adverse Outcome Pathways exemplified by linking decreased androgen receptor activity with decreased anogenital distance. Reproductive Toxicology, 128, 108662. https://doi.org/10.1016/j.reprotox.2024.108662
Leunbach, T. L., Berglund, A., Ernst, A., Hvistendahl, G. M., Rawashdeh, Y. F., & Gravholt, C. H. (2025). Prevalence, Incidence, and Age at Diagnosis of Boys With Hypospadias: A Nationwide Population-Based Epidemiological Study. Journal of Urology, 213(3), 350–360. https://doi.org/10.1097/JU.0000000000004319
Leung, J. K., & Sadar, M. D. (2017). Non-Genomic Actions of the Androgen Receptor in Prostate Cancer. Frontiers in Endocrinology, 8. https://doi.org/10.3389/fendo.2017.00002
Mattiske, D. M., & Pask, A. J. (2021). Endocrine disrupting chemicals in the pathogenesis of hypospadias; developmental and toxicological perspectives. Current Research in Toxicology, 2, 179–191. https://doi.org/10.1016/j.crtox.2021.03.004
OECD. (2016a). Test No. 421: Reproduction/Developmental Toxicity Screening Test. OECD. https://doi.org/10.1787/9789264264380-en
OECD. (2016b). Test No. 422: Combined Repeated Dose Toxicity Study with the Reproduction/Developmental Toxicity Screening Test. OECD. https://doi.org/10.1787/9789264264403-en
OECD. (2018a). Test No. 414: Prenatal Developmental Toxicity Study. OECD. https://doi.org/10.1787/9789264070820-en
OECD. (2018b). Test No. 443: Extended One-Generation Reproductive Toxicity Study. OECD. https://doi.org/10.1787/9789264185371-en
OECD. (2021). Test No. 416: Two-Generation Reproduction Toxicity (Section 4).
Paulozzi, L. J. (1999). International trends in rates of hypospadias and cryptorchidism.
Pedersen, E. B., Christiansen, S., & Svingen, T. (2022). AOP key event relationship report: Linking androgen receptor antagonism with nipple retention. Current Research in Toxicology, 3, 100085. https://doi.org/10.1016/j.crtox.2022.100085
Petiot, A., Perriton, C., Dickson, C., & Cohn, M. (2005). Development of the mammalian urethra is controlled by Fgfr2-IIIb. Development (Cambridge, England), 132(10), 2441–2450. https://doi.org/10.1242/dev.01778
Sinclair, A., Cao, M., Pask, A., Baskin, L., & Cunha, G. (2017). Flutamide-induced hypospadias in rats: A critical assessment. Differentiation; Research in Biological Diversity, 94, 37–57. https://doi.org/10.1016/j.diff.2016.12.001
Skakkebaek, N. E., Rajpert-De Meyts, E., Louis, G. M. B., Toppari, J., Andersson, A.-M., Eisenberg, M. L., Jensen, T. K., Jorgensen, N., Swan, S. H., Sapra, K. J., Ziebe, S., Priskorn, L., & Juul, A. (2016). Male Reproductive Disorders And Fertility Trends: Influences Of Environement And Genetic susceptibility. PHYSIOLOGICAL REVIEWS, 96(1), 55–97. https://doi.org/10.1152/physrev.00017.2015
Stewart, M. K., Mattiske, D. M., & Pask, A. J. (2018). In utero exposure to both high- and low-dose diethylstilbestrol disrupts mouse genital tubercle development†. Biology of Reproduction, 99(6), 1184–1193. https://doi.org/10.1093/biolre/ioy142
Svingen, T., Villeneuve, D. L., Knapen, D., Panagiotou, E. M., Draskau, M. K., Damdimopoulou, P., & O’Brien, J. M. (2021). A Pragmatic Approach to Adverse Outcome Pathway Development and Evaluation. Toxicological Sciences, 184(2), 183–190. https://doi.org/10.1093/toxsci/kfab113
Tut, T. G., Ghadessy, F. J., Trifiro, M. A., Pinsky, L., & Yong, E. L. (1997). Long Polyglutamine Tracts in the Androgen Receptor Are Associated with Reduced Trans -Activation, Impaired Sperm Production, and Male Infertility 1. The Journal of Clinical Endocrinology & Metabolism, 82(11), 3777–3782. https://doi.org/10.1210/jcem.82.11.4385
Welsh, M., Saunders, P. T. K., Fisken, M., Scott, H. M., Hutchison, G. R., Smith, L. B., & Sharpe, R. M. (2008). Identification in rats of a programming window for reproductive tract masculinization, disruption of which leads to hypospadias and cryptorchidism. Journal of Clinical Investigation, 118(4), 1479–1490. https://doi.org/10.1172/JCI34241
Willingham, E., Agras, K., Souza, A. J. de, Konijeti, R., Yucel, S., Rickie, W., Cunha, G., & Baskin, L. (2006). Steroid receptors and mammalian penile development: An unexpected role for progesterone receptor? The Journal of Urology, 176(2), 728–733. https://doi.org/10.1016/j.juro.2006.03.078
Yucel, S., Liu, W., Cordero, D., Donjacour, A., Cunha, G., & Baskin, L. (2004). Anatomical studies of the fibroblast growth factor-10 mutant, Sonic Hedge Hog mutant and androgen receptor mutant mouse genital tubercle. Advances in Experimental Medicine and Biology, 545, 123–148. https://doi.org/10.1007/978-1-4419-8995-6_8
Zheng, Z., Armfield, B., & Cohn, M. (2015). Timing of androgen receptor disruption and estrogen exposure underlies a spectrum of congenital penile anomalies. Proceedings of the National Academy of Sciences of the United States of America, 112(52), E7194-203. https://doi.org/10.1073/pnas.1515981112
Appendix 1
List of MIEs in this AOP
Event: 26: Antagonism, Androgen receptor
Short Name: Antagonism, Androgen receptor
Key Event Component
| Process | Object | Action |
|---|---|---|
| androgen receptor activity | androgen receptor | decreased |
AOPs Including This Key Event
| AOP ID and Name | Event Type |
|---|---|
| Aop:306 - Androgen receptor (AR) antagonism leading to short anogenital distance (AGD) in male (mammalian) offspring | MolecularInitiatingEvent |
| Aop:344 - Androgen receptor (AR) antagonism leading to nipple retention (NR) in male (mammalian) offspring | MolecularInitiatingEvent |
| Aop:345 - Androgen receptor (AR) antagonism leading to decreased fertility in females | MolecularInitiatingEvent |
| Aop:372 - Androgen receptor antagonism leading to testicular cancer | MolecularInitiatingEvent |
| Aop:477 - Androgen receptor (AR) antagonism leading to hypospadias in male (mammalian) offspring | MolecularInitiatingEvent |
| Aop:476 - Adverse Outcome Pathways diagram related to PBDEs associated male reproductive toxicity | MolecularInitiatingEvent |
| Aop:19 - Androgen receptor antagonism leading to adverse effects in the male foetus (mammals) | MolecularInitiatingEvent |
| Aop:595 - Nanoplastic effect | MolecularInitiatingEvent |
Stressors
| Name |
|---|
| Mercaptobenzole |
| Triticonazole |
| Flusilazole |
| Epoxiconazole |
| Prochloraz |
| Propiconazole |
| Tebuconazole |
| Flutamide |
| Cyproterone acetate |
| Vinclozolin |
Biological Context
| Level of Biological Organization |
|---|
| Molecular |
Cell term
| Cell term |
|---|
| eukaryotic cell |
Domain of Applicability
Taxonomic Applicability| Term | Scientific Term | Evidence | Links |
|---|---|---|---|
| mammals | mammals | High | NCBI |
| Life Stage | Evidence |
|---|---|
| During development and at adulthood | High |
| Sex | Evidence |
|---|---|
| Mixed | High |
Both the DNA-binding and ligand-binding domains of the AR are highly evolutionary conserved, whereas the transactivation domain show more divergence which may affect AR-mediated gene regulation across species (Davey & Grossmann, 2016). Despite certain inter-species differences, AR function mediated through gene expression is highly conserved, with mutations studies from both humans and rodents showing strong correlation for AR-dependent development and function (Walters et al, 2010).
This KE is applicable for both sexes, across developmental stages into adulthood, in numerous cells and tissues and across mammalian taxa. It is, however, acknowledged that this KE most likely has a much broader domain of applicability extending to non-mammalian vertebrates. AOP developers are encouraged to add additional relevant knowledge to expand on the applicability to also include other vertebrates.
Key Event Description
The androgen receptor (AR) and its function
The AR is a ligand-activated transcription factor belonging to the steroid hormone nuclear receptor family (Davey & Grossmann, 2016). The AR has three domains: the N-terminal domain, the DNA-binding domain and the ligand-binding domain, with the latter being most evolutionary conserved. Testosterone (T) and the more biologically active dihydrotestosterone (DHT) are endogenous ligands for the AR (MacLean et al, 1993; MacLeod et al, 2010; Schwartz et al, 2019). In teleost fishes, 11-ketotestosterone is the second main ligand (Schuppe et al, 2020). Human AR mutations and mouse knock-out models have established a pivotal role for the AR in masculinization and spermatogenesis (Walters et al, 2010). Apart from the essential role for AR in male reproductive development and function (Walters et al, 2010), the AR is also expressed in many other tissues and organs such as bone, muscles, ovaries, and the immune system (Rana et al, 2014).
AR antagonism as Key Event
The main function of the AR is to activate gene transcription in cells. Canonical signaling occurs by ligands (androgens) binding to AR in the cytoplasm which results in translocation to the cell nucleus, receptor dimerization and binding to specific regulatory DNA sequences (Heemers & Tindall, 2007). The gene targets regulated by AR activation depends on cell/tissue type and what stage of development activation occur, and is, for instance, dependent on available co-factors. Apart from the canonical signaling pathway, AR can also initiate cytoplasmic signaling pathways with other functions than the nuclear pathway, for instance rapid change in cell function by ion transport changes (Heinlein & Chang, 2002) and association with Src kinase to activate MAPK/ERK signaling and activation of the PI3K/Akt pathway (Leung & Sadar, 2017).
How it is Measured or Detected
AR antagonism can be measured in vitro by transient or stable transactivation assays to evaluate nuclear receptor activation. There is already a validated test guideline for AR (ant)agonism adopted by the OECD, Test No. 458: Stably Transfected Human Androgen Receptor Transcriptional Activation Assay for Detection of Androgenic Agonist and Antagonist Activity of Chemicals (OECD, 2016). This test guideline contains three different methods. More information on limitations, advantages, protocols, and availability and description of cells are given in the test guideline.
Besides these validated methods, other transiently or stably transfected reporter cell lines are available as well as yeast based systems (Campana et al, 2015; Körner et al, 2004). AR nuclear translocation can be monitored by various assays (Campana et al 2015), for example by monitoring fluorescent rat AR movement in living cells (Tyagi et al 2020), with several human AR translocation assays being commercially available; e.g. Fluorescent AR Nuclear Translocation Assay (tGFP-hAR/HEK293) or Human Androgen NHR Cell Based Antagonist Translocation LeadHunter Assay.
Additional information on AR interaction can be obtained employing competitive AR binding assays (Freyberger et al 2010, Shaw et al 2018), which can also inform on relative potency of the compounds, though not on downstream effect of the AR binding.
The recently developed AR dimerization assay provides an assay with an improved ability to measure potential stressor-mediated disruption of dimerization/activation (Lee et al, 2021).
References
Campana C, Pezzi V, Rainey WE (2015) Cell based assays for screening androgen receptor ligands. Semin Reprod Med 33: 225-234.
Freyberger A, Weimer M, Tran HS, Ahr HJ. Assessment of a recombinant androgen receptor binding assay: initial steps towards validation. Reprod Toxicol. 2010 Aug;30(1):2-8. doi: 10.1016/j.reprotox.2009.10.001. Epub 2009 Oct 13. PMID: 19833195.
OECD (2022). Test No. 251: Rapid Androgen Disruption Activity Reporter (RADAR) assay. Paris: OECD Publishing doi:10.1787/da264d82-en.
Shaw J, Leveridge M, Norling C, Karén J, Molina DM, O'Neill D, Dowling JE, Davey P, Cowan S, Dabrowski M, Main M, Gianni D. Determining direct binders of the Androgen Receptor using a high-throughput Cellular Thermal Shift Assay. Sci Rep. 2018 Jan 9;8(1):163. doi: 10.1038/s41598-017-18650-x. PMID: 29317749; PMCID: PMC5760633.
Tyagi RK, Lavrovsky Y, Ahn SC, Song CS, Chatterjee B, Roy AK (2000) Dynamics of intracellular movement and nucleocytoplasmic recycling of the ligand-activated androgen receptor in living cells. Mol Endocrinol 14: 1162-1174
List of Key Events in the AOP
Event: 1614: Decrease, androgen receptor activation
Short Name: Decrease, AR activation
Key Event Component
| Process | Object | Action |
|---|---|---|
| androgen receptor activity | androgen receptor | decreased |
AOPs Including This Key Event
Biological Context
| Level of Biological Organization |
|---|
| Tissue |
Domain of Applicability
Taxonomic Applicability| Term | Scientific Term | Evidence | Links |
|---|---|---|---|
| mammals | mammals | High | NCBI |
| Life Stage | Evidence |
|---|---|
| During development and at adulthood | High |
| Sex | Evidence |
|---|---|
| Mixed | High |
This KE is considered broadly applicable across mammalian taxa as all mammals express the AR in numerous cells and tissues where it regulates gene transcription required for developmental processes and functions. It is, however, acknowledged that this KE most likely has a much broader domain of applicability extending to non-mammalian vertebrates. AOP developers are encouraged to add additional relevant knowledge to expand on the applicability to also include other vertebrates.
Key Event Description
This KE refers to decreased activation of the androgen receptor (AR) as occurring in complex biological systems such as tissues and organs in vivo. It is thus considered distinct from KEs describing either blocking of AR or decreased androgen synthesis.
The AR is a nuclear transcription factor with canonical AR activation regulated by the binding of the androgens such as testosterone or dihydrotestosterone (DHT). Thus, AR activity can be decreased by reduced levels of steroidal ligands (testosterone, DHT) or the presence of compounds interfering with ligand binding to the receptor (Davey & Grossmann, 2016; Gao et al., 2005).
In the inactive state, AR is sequestered in the cytoplasm of cells by molecular chaperones. In the classical (genomic) AR signaling pathway, AR activation causes dissociation of the chaperones, AR dimerization and translocation to the nucleus to modulate gene expression. AR binds to the androgen response element (ARE) (Davey & Grossmann, 2016; Gao et al., 2005). Notably, for transcriptional regulation the AR is closely associated with other co-factors that may differ between cells, tissues and life stages. In this way, the functional consequence of AR activation is cell- and tissue-specific. This dependency on co-factors such as the SRC proteins also means that stressors affecting recruitment of co-activators to AR can result in decreased AR activity (Heinlein & Chang, 2002).
Ligand-bound AR may also associate with cytoplasmic and membrane-bound proteins to initiate cytoplasmic signaling pathways with other functions than the nuclear pathway. Non-genomic AR signaling includes association with Src kinase to activate MAPK/ERK signaling and activation of the PI3K/Akt pathway. Decreased AR activity may therefore be a decrease in the genomic and/or non-genomic AR signaling pathways (Leung & Sadar, 2017).
How it is Measured or Detected
This KE specifically focuses on decreased in vivo activation, with most methods that can be used to measure AR activity carried out in vitro. They provide indirect information about the KE and are described in lower tier MIE/KEs (see for example MIE/KE-26 for AR antagonism, KE-1690 for decreased T levels and KE-1613 for decreased dihydrotestosterone levels). Assays may in the future be developed to measure AR activation in mammalian organisms.
References
Davey, R. A., & Grossmann, M. (2016). Androgen Receptor Structure, Function and Biology: From Bench to Bedside. The Clinical Biochemist. Reviews, 37(1), 3–15.
Gao, W., Bohl, C. E., & Dalton, J. T. (2005). Chemistry and structural biology of androgen receptor. Chemical Reviews, 105(9), 3352–3370. https://doi.org/10.1021/cr020456u
Heinlein, C. A., & Chang, C. (2002). Androgen Receptor (AR) Coregulators: An Overview. https://academic.oup.com/edrv/article/23/2/175/2424160
Leung, J. K., & Sadar, M. D. (2017). Non-Genomic Actions of the Androgen Receptor in Prostate Cancer. Frontiers in Endocrinology, 8. https://doi.org/10.3389/fendo.2017.00002
OECD (2022). Test No. 251: Rapid Androgen Disruption Activity Reporter (RADAR) assay. Paris: OECD Publishing doi:10.1787/da264d82-en.
|
|
|
Event: 286: Altered, Transcription of genes by the androgen receptor
Short Name: Altered, Transcription of genes by the AR
Key Event Component
| Process | Object | Action |
|---|---|---|
| regulation of gene expression | androgen receptor | decreased |
AOPs Including This Key Event
Stressors
| Name |
|---|
| Bicalutamide |
| Cyproterone acetate |
| Epoxiconazole |
| Flutamide |
| Flusilazole |
| Prochloraz |
| Propiconazole |
| Stressor:286 Tebuconazole |
| Triticonazole |
| Vinclozalin |
Biological Context
| Level of Biological Organization |
|---|
| Tissue |
Domain of Applicability
Taxonomic Applicability| Term | Scientific Term | Evidence | Links |
|---|---|---|---|
| mammals | mammals | High | NCBI |
| Life Stage | Evidence |
|---|---|
| During development and at adulthood | High |
| Sex | Evidence |
|---|---|
| Mixed | High |
Both the DNA-binding and ligand-binding domains of the AR are highly evolutionary conserved, whereas the transactivation domain show more divergence, which may affect AR-mediated gene regulation across species (Davey and Grossmann 2016). Despite certain inter-species differences, AR function mediated through gene expression is highly conserved, with mutation studies from both humans and rodents showing strong correlation for AR-dependent development and function (Walters et al. 2010).
This KE is considered broadly applicable across mammalian taxa, sex and developmental stages, as all mammals express the AR in numerous cells and tissues where it regulates gene transcription required for developmental processes and function. It is, however, acknowledged that this KE most likely has a much broader domain of applicability extending to non-mammalian vertebrates. AOP developers are encouraged to add additional relevant knowledge to expand on the applicability to also include other vertebrates.
Key Event Description
This KE refers to transcription of genes by the androgen receptor (AR) as occurring in complex biological systems such as tissues and organs in vivo. Rather than measuring individual genes, this KE aims to capture patterns of effects at transcriptome level in specific target cells/tissues. In other words, it can be replaced by specific KEs for individual adverse outcomes as information becomes available, for example the transcriptional toxicity response in prostate tissue for AO: prostate cancer, perineum tissue for AO: reduced AGD, etc. AR regulates many genes that differ between tissues and life stages and, importantly, different gene transcripts within individual cells can go in either direction since AR can act as both transcriptional activator and suppressor. Thus, the ‘directionality’ of the KE cannot be either reduced or increased, but instead describe an altered transcriptome.
The Androgen Receptor and its function
The AR belongs to the steroid hormone nuclear receptor family. It is a ligand-activated transcription factor with three domains: the N-terminal domain, the DNA-binding domain, and the ligand-binding domain with the latter being the most evolutionary conserved (Davey and Grossmann 2016). Androgens (such as dihydrotestosterone and testosterone) are AR ligands and act by binding to the AR in androgen-responsive tissues (Davey and Grossmann 2016). Human AR mutations and mouse knockout models have established a fundamental role for AR in masculinization and spermatogenesis (Maclean et al.; Walters et al. 2010; Rana et al. 2014). The AR is also expressed in many other tissues such as bone, muscles, ovaries and within the immune system (Rana et al. 2014).
Altered transcription of genes by the AR as a Key Event
Upon activation by ligand-binding, the AR translocates from the cytoplasm to the cell nucleus, dimerizes, binds to androgen response elements in the DNA to modulate gene transcription (Davey and Grossmann 2016). The transcriptional targets vary between cells and tissues, as well as with developmental stages and is also dependent on available co-regulators (Bevan and Parker 1999; Heemers and Tindall 2007). It should also be mentioned that the AR can work in other ‘non-canonial’ ways such as non-genomic signaling, and ligand-independent activation (Davey & Grossmann, 2016; Estrada et al, 2003; Jin et al, 2013).
A large number of known, and proposed, target genes of AR canonical signaling have been identified by analysis of gene expression following treatments with AR agonists (Bolton et al. 2007; Ngan et al. 2009, Jin et al. 2013).
How it is Measured or Detected
Altered transcription of genes by the AR can be measured by measuring the transcription level of known downstream target genes by RT-qPCR or other transcription analyses approaches, e.g. transcriptomics.
Since this KE aims to capture AR-mediated transcriptional patterns of effect, downstream bioinformatics analyses will typically be required to identify and compare effect footprints. Clusters of genes can be statistically associated with, for example, biological process terms or gene ontology terms relevant for AR-mediated signaling. Large transcriptomics data repositories can be used to compare transcriptional patterns between chemicals, tissues, and species (e.g. TOXsIgN (Darde et al, 2018a; Darde et al, 2018b), comparisons can be made to identified sets of AR ‘biomarker’ genes (e.g. as done in (Rooney et al, 2018)), and various methods can be used e.g. connectivity mapping (Keenan et al, 2019).
References
Bevan C, Parker M (1999) The role of coactivators in steroid hormone action. Exp. Cell Res. 253:349–356
Bolton EC, So AY, Chaivorapol C, et al (2007) Cell- and gene-specific regulation of primary target genes by the androgen receptor. Genes Dev 21:2005–2017. doi: 10.1101/gad.1564207
Darde, T. A., Gaudriault, P., Beranger, R., Lancien, C., Caillarec-Joly, A., Sallou, O., et al. (2018a). TOXsIgN: a cross-species repository for toxicogenomic signatures. Bioinformatics 34, 2116–2122. doi:10.1093/bioinformatics/bty040.
Darde, T. A., Chalmel, F., and Svingen, T. (2018b). Exploiting advances in transcriptomics to improve on human-relevant toxicology. Curr. Opin. Toxicol. 11–12, 43–50. doi:10.1016/j.cotox.2019.02.001.
Davey RA, Grossmann M (2016) Androgen Receptor Structure, Function and Biology: From Bench to Bedside. Clin Biochem Rev 37:3–15
Estrada M, Espinosa A, Müller M, Jaimovich E (2003) Testosterone Stimulates Intracellular Calcium Release and Mitogen-Activated Protein Kinases Via a G Protein-Coupled Receptor in Skeletal Muscle Cells. Endocrinology 144:3586–3597. doi: 10.1210/en.2002-0164
Heemers H V., Tindall DJ (2007) Androgen receptor (AR) coregulators: A diversity of functions converging on and regulating the AR transcriptional complex. Endocr. Rev. 28:778–808
Jin, Hong Jian, Jung Kim, and Jindan Yu. 2013. “Androgen Receptor Genomic Regulation.” Translational Andrology and Urology 2(3):158–77. doi: 10.3978/j.issn.2223-4683.2013.09.01
Keenan, A. B., Wojciechowicz, M. L., Wang, Z., Jagodnik, K. M., Jenkins, S. L., Lachmann, A., et al. (2019). Connectivity Mapping: Methods and Applications. Annu. Rev. Biomed. Data Sci. 2, 69–92. doi:10.1146/ANNUREV-BIODATASCI-072018-021211.
Maclean HE, Chu S, Warne GL, Zajact JD Related Individuals with Different Androgen Receptor Gene Deletions
MacLeod DJ, Sharpe RM, Welsh M, et al (2010) Androgen action in the masculinization programming window and development of male reproductive organs. In: International Journal of Andrology. Blackwell Publishing Ltd, pp 279–287
Ngan S, Stronach EA, Photiou A, et al (2009) Microarray coupled to quantitative RT–PCR analysis of androgen-regulated genes in human LNCaP prostate cancer cells. Oncogene 28:2051–2063. doi: 10.1038/onc.2009.68
Rana K, Davey RA, Zajac JD (2014) Human androgen deficiency: Insights gained from androgen receptor knockout mouse models. Asian J. Androl. 16:169–177
Rooney, J. P., Chorley, B., Kleinstreuer, N., and Corton, J. C. (2018). Identification of Androgen Receptor Modulators in a Prostate Cancer Cell Line Microarray Compendium. Toxicol. Sci. 166, 146–162. doi:10.1093/TOXSCI/KFY187.
Walters KA, Simanainen U, Handelsman DJ (2010) Molecular insights into androgen actions in male and female reproductive function from androgen receptor knockout models. Hum Reprod Update 16:543–558. doi: 10.1093/humupd/dmq003
List of Adverse Outcomes in this AOP
Event: 2082: Hypospadias, increased
Short Name: Hypospadias
Key Event Component
| Process | Object | Action |
|---|---|---|
| embryonic organ development | penis | abnormal |
AOPs Including This Key Event
Biological Context
| Level of Biological Organization |
|---|
| Organ |
Organ term
| Organ term |
|---|
| penis |
Domain of Applicability
Taxonomic Applicability| Term | Scientific Term | Evidence | Links |
|---|---|---|---|
| human | Homo sapiens | High | NCBI |
| mouse | Mus musculus | High | NCBI |
| rat | Rattus norvegicus | High | NCBI |
| mammals | mammals | NCBI |
| Life Stage | Evidence |
|---|---|
| Perinatal | High |
| Sex | Evidence |
|---|---|
| Male | High |
Taxonomic applicability: Numerous studies have shown an association in humans between in utero exposure to endocrine disrupting chemicals and hypospadias. In mice and rats, in utero exposure to several endocrine disrupting chemicals, in particular estrogens and antiandrogens, have been shown to cause hypospadias in male offspring at different frequencies (Mattiske & Pask, 2021). Androgen-driven development of the male external genitalia is evolutionary conserved in most mammals and, to some extent, also in other vertebrate classes (Gredler et al., 2014). Hypospadias can in principle occur in all animals that form a genital tubercle and have been observed in many domestic animal species and wildlife species.
Life stage applicability: Penis development is finished prenatally in humans, and hypospadias is diagnosed at birth (Baskin & Ebbers, 2006). In rodents, penis development is not fully completed until weeks after birth, but hypospadias may be identified in early postnatal life as well, and in some cases in late gestation (Sinclair et al., 2017).
Sex applicability: Hypospadias is primarily used in reference to malformation of the male external genitalia.
Key Event Description
Hypospadias is a malformation of the penis where the urethral opening is displaced from the tip of the glans, usually on the ventral side on the phallus. Most cases of hypospadias are milder where the urethral opening still appears on the glans proper or on the most distal part of the shaft. In more severe cases, the opening may be more proximally placed on the shaft or even as low as the scrotum or the perineum.
In addition to the misplacement of the urethral opening, hypospadias is associated with an absence of ventral prepuce, an excess of dorsal preputial tissue, and in some cases a downward curvature of the penis (chordee). Patients with hypospadias may need surgical repairment depending on severity, with more proximal hypospadias patients in most need of surgeries to achieve optimal functional and cosmetic results (Baskin, 2000; Baskin & Ebbers, 2006; Mattiske & Pask, 2021). The incidence of hypospadias varies greatly between countries, from 1:100 to 1:500 of newborn boys (Skakkebaek et al., 2016), and the global prevalence seems to be increasing (Paulozzi, 1999; Springer et al., 2016; Yu et al., 2019).
The external genitalia arise from the biphasic genital tubercle during fetal development. Androgens (testosterone and dihydrotestosterone) drive formation of the male external genitalia. In humans, the urethra develops by fusion of two endoderm-derived urethral folds. Disruption of genital tubercle differentiation results in an incomplete urethra, i.e. hypospadias. (Baskin, 2000; Baskin & Ebbers, 2006).
How it is Measured or Detected
In humans, hypospadias is diagnosed clinically by physical examination of the infant and is at first recognized by the absence of ventral prepuce and concurrent excess dorsal prepuce (Baskin, 2000). Hypospadias may be classified according to the location of the urethral meatus: Glandular, subcoronal, midshaft, penoscrotal, scrotal, and perineal (Baskin & Ebbers, 2006).
In mice and rats, macroscopic assessment of hypospadias may be performed postnatally, and several OECD test guidelines require macroscopic examination of genital abnormalities in in vivo toxicity studies (TG 414, 416, 421/422, 443). The guidelines do not define hypospadias or how to identify them. Fetal and neonatal identification of hypospadias may require microscopic examination for proper evaluation of the pathology. This can be done by scanning electron microscopy (Uda et al., 2004), or by histological assessment in which the presence of the urethral opening in proximal, transverse sections (for example co-occuring with the os penis or corpus cavernosum), indicates hypospadias (Mahawong et al., 2014; Sinclair et al., 2017; Vilela et al., 2007). In a semiquantitative, histologic approach, the number of transverse sections of the penis with internalization of the urethra was related to the total length of the penis, achieving a percentage of urethral internalization. In this study, ≤89% of urethral internalization was defined as indicative of mild hypospadias (Stewart et al., 2018).
Regulatory Significance of the AO
In the OECD guidelines for developmental and reproductive toxicology, several test endpoints include examination of structural abnormalities with special attention to the organs of the reproductive system. These are: Test No. 414 ‘Prenatal Developmental Toxicity Study’ (OECD, 2018a); Test No. 416 ‘Two-Generation Reproduction Toxicity’ (OECD, 2001) and Tests No. 421/422 ‘Reproduction/Developmental Toxicity Screening Test’ (OECD, 2016a, 2016b). In Test No. 443 ‘Extended One-Generation Reproductive Toxicity Study’ (OECD, 2018b), hypospadias is specifically mentioned as a genital abnormality to note.
References
Baskin, L. S. (2000). Hypospadias and urethral development. The Journal of Urology, 163(3), 951–956.
Baskin, L. S., & Ebbers, M. B. (2006). Hypospadias: Anatomy, etiology, and technique. Journal of Pediatric Surgery, 41(3), 463–472. https://doi.org/10.1016/j.jpedsurg.2005.11.059
Gredler, M. L., Larkins, C. E., Leal, F., Lewis, A. K., Herrera, A. M., Perriton, C. L., Sanger, T. J., & Cohn, M. J. (2014). Evolution of External Genitalia: Insights from Reptilian Development. Sexual Development, 8(5), 311–326. https://doi.org/10.1159/000365771
Mahawong, P., Sinclair, A., Li, Y., Schlomer, B., Rodriguez, E., Ferretti, M. M., Liu, B., Baskin, L. S., & Cunha, G. R. (2014). Prenatal diethylstilbestrol induces malformation of the external genitalia of male and female mice and persistent second-generation developmental abnormalities of the external genitalia in two mouse strains. Differentiation, 88(2–3), 51–69. https://doi.org/10.1016/j.diff.2014.09.005
Mattiske, D. M., & Pask, A. J. (2021). Endocrine disrupting chemicals in the pathogenesis of hypospadias; developmental and toxicological perspectives. Current Research in Toxicology, 2, 179–191. https://doi.org/10.1016/j.crtox.2021.03.004
OECD. (2001). Test No. 416: Two-Generation Reproduction Toxicity. In OECD Guidelines for the Testing of Chemicals, Section 4. OECD Publishing. https://doi.org/10.1787/9789264070868-en
OECD. (2018). Test No. 414: Prenatal Developmental Toxicity Study. In OECD Guidelines for the Testing of Chemicals, Section 4. OECD Publishing. https://doi.org/10.1787/9789264070820-en
OECD. (2025a). Test No. 421: Reproduction/Developmental Toxicity Screening Test. In OECD Guidelines for the Testing of Chemicals, Section 4. OECD Publishing. https://doi.org/doi.org/10.1787/9789264264380-en
OECD. (2025b). Test No. 422: Combined Repeated Dose Toxicity Study with the Reproduction/Developmental Toxicity Screening Test. In OECD Guidelines for the Testing of Chemicals, Section 4. OECD Publising. https://doi.org/doi.org/10.1787/9789264264403-en
OECD. (2025c). Test No. 443: Extended One-Generation Reproductive Toxicity Study. In OECD Guidelines for the Testing of Chemicals, Section 4. OECD Publishing. https://doi.org/doi.org/10.1787/9789264185371-en
Paulozzi, L. J. (1999). International Trends in Rates of Hypospadias and Cryptorchidism. Environmental Health Perspectives, 107(4), 297–302. https://doi.org/10.1289/ehp.99107297
Sinclair, A. W., Cao, M., Pask, A., Baskin, L., & Cunha, G. R. (2017). Flutamide-induced hypospadias in rats: A critical assessment. Differentiation, 94, 37–57. https://doi.org/10.1016/j.diff.2016.12.001
Skakkebaek, N. E., Rajpert-De Meyts, E., Buck Louis, G. M., Toppari, J., Andersson, A.-M., Eisenberg, M. L., Jensen, T. K., Jørgensen, N., Swan, S. H., Sapra, K. J., Ziebe, S., Priskorn, L., & Juul, A. (2016). Male Reproductive Disorders and Fertility Trends: Influences of Environment and Genetic Susceptibility. Physiological Reviews, 96(1), 55–97. https://doi.org/10.1152/physrev.00017.2015.-It
Springer, A., van den Heijkant, M., & Baumann, S. (2016). Worldwide prevalence of hypospadias. Journal of Pediatric Urology, 12(3), 152.e1-152.e7. https://doi.org/10.1016/j.jpurol.2015.12.002
Stewart, M. K., Mattiske, D. M., & Pask, A. J. (2018). In utero exposure to both high- and low-dose diethylstilbestrol disrupts mouse genital tubercle development. Biology of Reproduction, 99(6), 1184–1193. https://doi.org/10.1093/biolre/ioy142
Uda, A., Kojima, Y., Hayashi, Y., Mizuno, K., Asai, N., & Kohri, K. (2004). Morphological features of external genitalia in hypospadiac rat model: 3-dimensional analysis. The Journal of Urology, 171(3), 1362–1366. https://doi.org/10.1097/01.JU.0000100140.42618.54
Vilela, M. L. B., Willingham, E., Buckley, J., Liu, B. C., Agras, K., Shiroyanagi, Y., & Baskin, L. S. (2007). Endocrine Disruptors and Hypospadias: Role of Genistein and the Fungicide Vinclozolin. Urology, 70(3), 618–621. https://doi.org/10.1016/j.urology.2007.05.004
Yu, X., Nassar, N., Mastroiacovo, P., Canfield, M., Groisman, B., Bermejo-Sánchez, E., Ritvanen, A., Kiuru-Kuhlefelt, S., Benavides, A., Sipek, A., Pierini, A., Bianchi, F., Källén, K., Gatt, M., Morgan, M., Tucker, D., Canessa, M. A., Gajardo, R., Mutchinick, O. M., … Agopian, A. J. (2019). Hypospadias Prevalence and Trends in International Birth Defect Surveillance Systems, 1980–2010. European Urology, 76(4), 482–490. https://doi.org/10.1016/j.eururo.2019.06.027
Appendix 2
List of Key Event Relationships in the AOP
List of Adjacent Key Event Relationships
Relationship: 2130: Antagonism, Androgen receptor leads to Decrease, AR activation
AOPs Referencing Relationship
| AOP Name | Adjacency | Weight of Evidence | Quantitative Understanding |
|---|---|---|---|
| Androgen receptor (AR) antagonism leading to short anogenital distance (AGD) in male (mammalian) offspring | adjacent | High | High |
| Androgen receptor (AR) antagonism leading to nipple retention (NR) in male (mammalian) offspring | adjacent | High | |
| Androgen receptor (AR) antagonism leading to hypospadias in male (mammalian) offspring | adjacent | High | |
| Androgen receptor (AR) antagonism leading to decreased fertility in females | adjacent | High | Moderate |
Evidence Supporting Applicability of this Relationship
| Term | Scientific Term | Evidence | Links |
|---|---|---|---|
| mammals | mammals | High | NCBI |
| Life Stage | Evidence |
|---|---|
| During development and at adulthood | High |
| Sex | Evidence |
|---|---|
| Mixed | High |
This KER is applicable to mammals as AR expression and activity is highly conserved (Davey & Grossmann, 2016). AR activity is important for sexual development and reproduction in both males and females (Prizant et al., 2014; Walters et al., 2010). AR function is required during development, puberty, and adulthood. It is, however, acknowledged that this KER most likely has a much broader domain of applicability extending to non-mammalian vertebrates. AOP developers are encouraged to add additional relevant knowledge to expand on the applicability to also include other vertebrates.
Key Event Relationship Description
The androgen receptor (AR) is a ligand-activated steroid hormone nuclear receptor (Davey & Grossmann, 2016). In its inactive state, the AR locates to the cytoplasm (Roy et al., 2001). When activated, the AR translocates to the nucleus, dimerizes, and, together with co-regulators, binds to specific DNA regulatory sequences to regulate gene transcription (Davey & Grossmann, 2016) (Lamont and Tindall, 2010). This is considered the canonical AR signaling pathway. The AR can also activate non-genomic signalling (Jin et al., 2013). However, this KER focuses on the canonical pathway.
The two main AR ligands are the androgens testosterone (T) and the more potent dihydrotestosterone (DHT). Androgens bind to the AR to mediate downstream androgenic responses, such as male development and masculinization (Rey, 2021; Walters et al., 2010). Antagonism of the AR would decrease AR activation and therefore the downstream AR-mediated effects.
Evidence Supporting this KER
Biological PlausibilityThe biological plausibility for this KER is considered high.
The AR belongs to the steroid hormone nuclear receptor family. The AR has 3 main domains essential for its activity, the N-terminal domain, the ligand binding domain, and the DNA binding domain (Roy et al., 2001). Ligands, such as T and DHT, must bind to the ligand binding domain to activate AR allowing it to fulfill its role as a transcription factor. The binding of the ligand induces a change in AR conformation allowing it to translocate to the nucleus and congregate into a subnuclear compartment (Marcelli et al., 2006; Roy et al., 2001) homodimerize and bind to the DNA target sequences and regulate transcription of target genes. Regulation of AR target genes is greatly facilitated by numerous co-factors. Active AR signaling is essential for male reproduction and sexual development and is also crucial in several other tissues and organs such as ovaries, the immune system, bones, and muscles (Ogino et al., 2011; Prizant et al., 2014; Rey, 2021; William H. Walker, 2021).
AR antagonists can compete with or prevent in different ways AR ligand binding, thereby preventing AR activation. Antagonism of the AR can prevent translocation to the nucleus, compartmentalization, dimerization and DNA binding. Consequently, AR cannot regulate transcription of target genes and androgen signalling is disrupted. This can be observed using different AR activation assays such as AR dimerization, translocation, DNA binding or transcriptional activity assays (Brown et al., 2023; OECD, 2020).
Empirical EvidenceThe empirical evidence for this KER is considered high
The effects of AR antagonism have been shown in many studies in vivo and in vitro.
Several stressors can act as antagonists of the AR and lead to decreased AR activation. Some of these are detailed in an AOP key event relationship report by (Pedersen et al., 2022) and shown below, exhibiting evidence of dose-concordance:
Stressors
- Cyproterone acetate: Using the AR-CALUX reporter assay in antagonism mode, cyproterone acetate showed an IC50 of 7.1 nM (Sonneveld, 2005)
- Epoxiconazole: Using transiently AR-transfected CHO cells, epoxiconazole showed a LOEC of 1.6 µM and an IC50 of 10 µM (Kjærstad et al., 2010).
- Flutamide: Using the AR-CALUX reporter assay in antagonism mode, flutamide showed an IC50 of 1.3 µM (Sonneveld, 2005).
- Flusilazole: Using hAR-EcoScreen Assay, triticonazole showed a LOEC for antagonisms of 0.8 µM and an IC50 of 2.8 (±0.1) µM (Draskau et al., 2019).
- Prochloraz: Using transiently AR-transfected CHO cells, prochloraz showed a LOEC of 6.3 µM and an IC50 of 13 µM (Kjærstad et al., 2010).
- Propiconazole: Using transiently AR-transfected CHO cells, propiconazole showed a LOEC of 12.5 µM and an IC50 of 18 µM (Kjærstad et al., 2010).
- Tebuconazole: Using transiently AR-transfected CHO cells, tebuconazole showed a LOEC of 3.1 µM and an IC50 of 8.1 µM (Kjærstad et al., 2010).
- Triticonazole: Using hAR-EcoScreen Assay, triticonazole showed a LOEC for antagonisms of 0.2 µM and an IC50 of 0.3 (±0.01) µM (Draskau et al., 2019).
- Vinclozolin: Using the AR-CALUX reporter assay in antagonism mode, vinclozolin showed an IC50of 1.0 µM(Sonneveld, 2005).”(Pedersen et al., 2022)
Other evidence:
Known AR antagonists are used for treatment of AR-sensitive cancers such as flutamide for prostate cancer (Mahler et al., 1998).
Uncertainties and Inconsistencies
Known antiandrogenic compounds like hydroxyflutamide have been shown to act as agonists when the AR carries certain mutations, therefore contributing to uncertainties (Yeh et al., 1997). Additionally, the levels of endogenous androgens (e.g., testosterone or dihydrotestosterone) and the variability in the presence and function of AR co-activators may modulate the effect of AR antagonism.
Quantitative Understanding of the Linkage
Response-response relationshipThe quantitative relationship between AR antagonism and AR activation will depend on the type of antagonist.
Time-scaleNuclear translocation in HeLa cells transfected with AR-GFP show a response within 2 hours after ligand exposure (Marcelli et al., 2006; Szafran et al., 2008). Another assay focusing on AR binding to promoters in LNCaP cells has shown that after ligand binding, AR is able to translocate and bind to the DNA sequences within 15min showing the speed of AR activation (Kang et al., 2002).
Known Feedforward/Feedback loops influencing this KERAR antagonism can lead to increased AR transcript stability and levels as a compensatory mechanism in prostate cancer cells (Dart et al., 2020). In turn, in presence of increased AR levels, AR antagonists can exhibit agonistic activity (Chen et al., 2003).
References
Brown, E. C., Hallinger, D. R., Simmons, S. O., Puig-Castellví, F., Eilebrecht, E., Arnold, L., & Bioscience, P. A. (2023). High-throughput AR dimerization assay identifies androgen disrupting chemicals and metabolites. Front. Toxicol, 5, 1134783. https://doi.org/10.3389/ftox.2023.1134783
Chen, C. D., Welsbie, D. S., Tran, C., Baek, S. H., Chen, R., Vessella, R., Rosenfeld, M. G., & Sawyers, C. L. (2003). A R T I C L E S Molecular determinants of resistance to antiandrogen therapy. NATURE MEDICINE, 10(1). https://doi.org/10.1038/nm972
Dart, D. A., Ashelford, K., & Jiang, W. G. (2020). AR mRNA stability is increased with AR-antagonist resistance via 3′UTR variants. https://doi.org/10.1530/EC-19-0340
Davey, R. A., & Grossmann, M. (2016). Androgen Receptor Structure, Function and Biology: From Bench to Bedside. In Androgen Receptor Biology Clin Biochem Rev (Vol. 37, Issue 1).
Draskau, M. K., Boberg, J., Taxvig, C., Pedersen, M., Frandsen, H. L., Christiansen, S., & Svingen, T. (2019). In vitro and in vivo endocrine disrupting effects of the azole fungicides triticonazole and flusilazole. Environmental Pollution, 255, 113309. https://doi.org/10.1016/j.envpol.2019.113309
Jin, H. J., Kim, J., & Yu, J. (2013). Androgen receptor genomic regulation. In Translational Andrology and Urology (Vol. 2, Issue 3, pp. 158–177). AME Publishing Company. https://doi.org/10.3978/j.issn.2223-4683.2013.09.01
Kang, Z., Pirskanen, A., Jänne, O. A., & Palvimo, J. J. (2002). Involvement of Proteasome in the Dynamic Assembly of the Androgen Receptor Transcription Complex. Journal of Biological Chemistry, 277(50), 48366–48371. https://doi.org/10.1074/jbc.M209074200
Kjærstad, M. B., Taxvig, C., Nellemann, C., Vinggaard, A. M., & Andersen, H. R. (2010). Endocrine disrupting effects in vitro of conazole antifungals used as pesticides and pharmaceuticals. Reproductive Toxicology, 30(4), 573–582. https://doi.org/10.1016/j.reprotox.2010.07.009
Lamont, K. R., and Tindall, D. J. (2010). Androgen Regulation of Gene Expression. Adv. Cancer Res. 107, 137–162. doi:10.1016/S0065-230X(10)07005-3.
Mahler, C., Verhelst, J., and Denis, L. (1998). Clinical pharmacokinetics of the antiandrogens and their efficacy in prostate cancer. Clin. Pharmacokinet. 34, 405–417. doi:10.2165/00003088-199834050-00005/METRICS.
Marcelli, M., Stenoien, D. L., Szafran, A. T., Simeoni, S., Agoulnik, I. U., Weigel, N. L., Moran, T., Mikic, I., Price, J. H., & Mancini, M. A. (2006). Quantifying effects of ligands on androgen receptor nuclear translocation, intranuclear dynamics, and solubility. Journal of Cellular Biochemistry, 98(4), 770–788. https://doi.org/10.1002/jcb.20593
OECD (2020). Test No. 458: Stably Transfected Human Androgen Receptor Transcriptional Activation Assay for Detection of Androgenic Agonist and Antagonist Activity of Chemicals. OECD Guide. Paris: OECD Publishing doi:10.1787/9789264264366-en.
Ogino, Y., Miyagawa, S., Katoh, H., Prins, G. S., Iguchi, T., & Yamada, G. (2011). Essential functions of androgen signaling emerged through the developmental analysis of vertebrate sex characteristics. Evolution & Development, 13(3), 315–325. https://doi.org/10.1111/j.1525-142X.2011.00482.x
Pedersen, E. B., Christiansen, S., & Svingen, T. (2022). AOP key event relationship report: Linking androgen receptor antagonism with nipple retention. Current Research in Toxicology, 3, 100085. https://doi.org/10.1016/j.crtox.2022.100085
Prizant, H., Gleicher, N., & Sen, A. (2014). Androgen actions in the ovary: balance is key. Journal of Endocrinology, 222(3), R141–R151. https://doi.org/10.1530/JOE-14-0296
Rey, R. A. (2021). The Role of Androgen Signaling in Male Sexual Development at Puberty. Endocrinology, 162(2). https://doi.org/10.1210/endocr/bqaa215
Roy, A. K., Tyagi, R. K., Song, C. S., Lavrovsky, Y., Ahn, S. C., Oh, T. S., & Chatterjee, B. (2001). Androgen receptor: Structural domains and functional dynamics after ligand-receptor interaction. Annals of the New York Academy of Sciences, 949, 44–57. https://doi.org/10.1111/j.1749-6632.2001.tb04001.x
Sonneveld, E. (2005). Development of Androgen- and Estrogen-Responsive Bioassays, Members of a Panel of Human Cell Line-Based Highly Selective Steroid-Responsive Bioassays. Toxicological Sciences, 83(1), 136–148. https://doi.org/10.1093/toxsci/kfi005
Szafran, A. T., Szwarc, M., Marcelli, M., & Mancini, M. A. (2008). Androgen Receptor Functional Analyses by High Throughput Imaging: Determination of Ligand, Cell Cycle, and Mutation-Specific Effects. PLoS ONE, 3(11), e3605. https://doi.org/10.1371/journal.pone.0003605
Walters, K. A., Simanainen, U., & Handelsman, D. J. (2010). Molecular insights into androgen actions in male and female reproductive function from androgen receptor knockout models. In Human Reproduction Update (Vol. 16, Issue 5, pp. 543–558). Hum Reprod Update. https://doi.org/10.1093/humupd/dmq003
William H. Walker. (2021). Androgen Actions in the Testis and the Regulation of Spermatogenesis. In Advances in Experimental Medicine and Biology: Vol. volume 1381 (pp. 175–203).
Yeh, S., Miyamoto, H., & Chang, C. (1997). ARA70 and androgenic activity of hydroxyflutamide Hydroxyflutamide may not always be a pure antiandrogen (Vol. 349).
Relationship: 2124: Decrease, AR activation leads to Altered, Transcription of genes by the AR
AOPs Referencing Relationship
Evidence Supporting Applicability of this Relationship
| Term | Scientific Term | Evidence | Links |
|---|---|---|---|
| mammals | mammals | High | NCBI |
| Life Stage | Evidence |
|---|---|
| During development and at adulthood | High |
| Sex | Evidence |
|---|---|
| Mixed | High |
This KER is applicable for both sexes, across developmental stages into adulthood, in numerous cells and tissues and across mammalian taxa. It is, however, acknowledged that this KER most likely has a much broader domain of applicability extending to non-mammalian vertebrates. AOP developers are encouraged to add additional relevant knowledge to expand on the applicability to also include other vertebrates.
Key Event Relationship Description
The androgen receptor (AR) is a ligand-dependent nuclear transcription factor that upon activation translocates to the nucleus, dimerizes, and binds androgen response elements (AREs) to modulate transcription of target genes (Lamont and Tindall, 2010, Roy et al. 2001). Decreased activation of the AR affects its transcription factor activity, therefore leading to altered AR-target gene expression. This KER refers to decreased AR activation and altered gene expression occurring in complex systems, such as in vivo and the specific effect on transcription of AR target genes will depend on species, life stage, tissue, cell type etc.
Evidence Supporting this KER
Biological PlausibilityThe biological plausibility for this KER is considered high
The AR is a ligand-activated transcription factor part of the steroid hormone nuclear receptor family. Non-activated AR is found in the cytoplasm as a multiprotein complex with heat-shock proteins, immunophilins and, other chaperones (Roy et al. 2001). Upon activation through ligand binding, the AR dissociates from the protein complex, translocates to the nucleus and homodimerizes. Facilitated by co-regulators, AR can bind to DNA regions containing AREs and initiate transcription of target genes, that thus will be different in e.g. different tissues, life-stages, species etc.
Through mapping of AREs and ChIP sequencing studies, several AR target genes have been identified, mainly studied in prostate cells (Jin, Kim, and Yu 2013). Different co-regulators and ligands lead to altered expression of different sets of genes (Jin et al. 2013; Kanno et al. 2022). Alternative splicing of the AR can lead to different AR variants that also affects which genes are transcribed (Jin et al. 2013).
Apart from this canonical signaling pathway, the AR can suppress gene expression, indirectly regulate miRNA transcription, and have non-genomic effects by rapid activation of second messenger pathways in either presence or absence of a ligand (Jin et al. 2013).
Empirical EvidenceThe empirical evidence for this KER is considered high
In humans, altered gene expression profiling in individuals with androgen insensitivity syndrome (AIS) can provide supporting empirical evidence (Holterhus et al. 2003; Peng et al. 2021). In rodent AR knockout (KO) models, gene expression profiling studies and gene-targeted approaches have provided information on differentially expressed genes in several organ systems including male and female reproductive, endocrine, muscular, cardiovascular and nervous systems (Denolet et al. 2006; Fan et al. 2005; Holterhus et al. 2003; Ikeda et al. 2005; Karlsson et al. 2016; MacLean et al. 2008; Rana et al. 2011; Russell et al. 2012; Shiina et al. 2006; Wang et al. 2006; Welsh et al. 2012; Willems et al. 2010; Yu et al. 2008, 2012; Zhang et al. 2006; Zhou et al. 2011).
Exposure to known antiandrogens has been shown to alter transcriptional profiles, for example of neonatal pig ovaries (Knapczyk-Stwora et al. 2019).
Dose concordance has also been observed for instance in zebrafish embryos; a dose of 50 µg/L of the AR antagonist flutamide resulted in 674 differentially expressed genes at 96 h post fertilization whereas 500 µg/L flutamide resulted in 2871 differentially expressed genes (Ayobahan et al., 2023).
Uncertainties and InconsistenciesAR action has been reported to occur also without ligand binding. However, not much is known about the extent and biological implications of such non-canonical, ligand-independent AR activation (Bennesch and Picard 2015).
Quantitative Understanding of the Linkage
Response-response relationshipThere is not enough data to define a quantitative relationship between AR activation and alteration of AR target gene transcription, and such a relationship will differ between biological systems (species, tissue, cell type, life stage etc).
Time-scaleAR and promoter interactions occur within 15 minutes of ligand binding, RNA polymerase II and coactivator recruitment are proposed to occur transiently with cycles of approximately 90 minutes in LNCaP cells (Kang et al. 2002). RNA polymerase II elongation rates in mammalian cells have been shown to range between 1.3 and 4.3 kb/min (Maiuri et al. 2011). Therefore, depending on the cell type and the half-life of the AR target gene transcripts, changes are to be expected within hours.
Known modulating factors| Modulating Factor (MF) | MF Specification | Effect(s) on the KER | Reference(s) |
|---|---|---|---|
| Age | AR expression in aging male rats | Tissue-specific alterations in AR activity with aging | (Supakar et al. 1993; Wu, Lin, and Gore 2009) |
| Genotype | Number of CAG repeats in the first exon of AR | Decreased AR activation with increased number of CAGs |
(Tut et al. 1997; Chamberlain et al. 1994) |
AR has been hypothesized to auto-regulate its mRNA and protein levels (Mora and Mahesh 1999).
References
Ayobahan, S. U., Alvincz, J., Reinwald, H., Strompen, J., Salinas, G., Schäfers, C., et al. (2023). Comprehensive identification of gene expression fingerprints and biomarkers of sexual endocrine disruption in zebrafish embryo. Ecotoxicol. Environ. Saf. 250, 114514. doi:10.1016/J.ECOENV.2023.114514.
Bennesch, Marcela A., and Didier Picard. 2015. “Minireview: Tipping the Balance: Ligand-Independent Activation of Steroid Receptors.” Molecular Endocrinology 29(3):349–63.
Chamberlain, Nancy L., Erika D. Driverand, and Roger L. Miesfeldi. 1994. The Length and Location of CAG Trinucleotide Repeats in the Androgen Receptor N-Terminal Domain Affect Transactivation Function. Vol. 22.
Denolet, Evi, Karel De Gendt, Joke Allemeersch, Kristof Engelen, Kathleen Marchal, Paul Van Hummelen, Karen A. L. Tan, Richard M. Sharpe, Philippa T. K. Saunders, Johannes V. Swinnen, and Guido Verhoeven. 2006. “The Effect of a Sertoli Cell-Selective Knockout of the Androgen Receptor on Testicular Gene Expression in Prepubertal Mice.” Molecular Endocrinology 20(2):321–34. doi: 10.1210/me.2005-0113.
Fan, Wuqiang, Toshihiko Yanase, Masatoshi Nomura, Taijiro Okabe, Kiminobu Goto, Takashi Sato, Hirotaka Kawano, Shigeaki Kato, and Hajime Nawata. 2005. Androgen Receptor Null Male Mice Develop Late-Onset Obesity Caused by Decreased Energy Expenditure and Lipolytic Activity but Show Normal Insulin Sensitivity With High Adiponectin Secretion. Vol. 54.
Holterhus, Paul-Martin, Olaf Hiort, Janos Demeter, Patrick O. Brown, and James D. Brooks. 2003. Differential Gene-Expression Patterns in Genital Fibroblasts of Normal Males and 46,XY Females with Androgen Insensitivity Syndrome: Evidence for Early Programming Involving the Androgen Receptor. Vol. 4.
Ikeda, Yasumasa, Ken Ichi Aihara, Takashi Sato, Masashi Akaike, Masanori Yoshizumi, Yuki Suzaki, Yuki Izawa, Mitsunori Fujimura, Shunji Hashizume, Midori Kato, Shusuke Yagi, Toshiaki Tamaki, Hirotaka Kawano, Takahiro Matsumoto, Hiroyuki Azuma, Shigeaki Kato, and Toshio Matsumoto. 2005. “Androgen Receptor Gene Knockout Male Mice Exhibit Impaired Cardiac Growth and Exacerbation of Angiotensin II-Induced Cardiac Fibrosis.” Journal of Biological Chemistry 280(33):29661–66. doi: 10.1074/jbc.M411694200.
Jin, Hong Jian, Jung Kim, and Jindan Yu. 2013. “Androgen Receptor Genomic Regulation.” Translational Andrology and Urology 2(3):158–77.
Kang, Zhigang, Asta Pirskanen, Olli A. Jänne, and Jorma J. Palvimo. 2002. “Involvement of Proteasome in the Dynamic Assembly of the Androgen Receptor Transcription Complex.” Journal of Biological Chemistry 277(50):48366–71. doi: 10.1074/jbc.M209074200.
Kanno, Yuichiro, Nao Saito, Ryota Saito, Tomohiro Kosuge, Ryota Shizu, Tomofumi Yatsu, Takuomi Hosaka, Kiyomitsu Nemoto, Keisuke Kato, and Kouichi Yoshinari. 2022. “Differential DNA-Binding and Cofactor Recruitment Are Possible Determinants of the Synthetic Steroid YK11-Dependent Gene Expression by Androgen Receptor in Breast Cancer MDA-MB 453 Cells.” Experimental Cell Research 419(2). doi: 10.1016/j.yexcr.2022.113333.
Karlsson, Sara A., Erik Studer, Petronella Kettunen, and Lars Westberg. 2016. “Neural Androgen Receptors Modulate Gene Expression and Social Recognition but Not Social Investigation.” Frontiers in Behavioral Neuroscience 10(MAR). doi: 10.3389/fnbeh.2016.00041.
Knapczyk-Stwora, Katarzyna, Anna Nynca, Renata E. Ciereszko, Lukasz Paukszto, Jan P. Jastrzebski, Elzbieta Czaja, Patrycja Witek, Marek Koziorowski, and Maria Slomczynska. 2019. “Flutamide-Induced Alterations in Transcriptional Profiling of Neonatal Porcine Ovaries.” Journal of Animal Science and Biotechnology 10(1):1–15. doi: 10.1186/s40104-019-0340-y.
Lamont, K. R., and Tindall, D. J. (2010). Androgen Regulation of Gene Expression. Adv. Cancer Res. 107, 137–162. doi:10.1016/S0065-230X(10)07005-3.
MacLean, Helen E., W. S. Maria Chiu, Amanda J. Notini, Anna-Maree Axell, Rachel A. Davey, Julie F. McManus, Cathy Ma, David R. Plant, Gordon S. Lynch, and Jeffrey D. Zajac. 2008. “ Impaired Skeletal Muscle Development and Function in Male, but Not Female, Genomic Androgen Receptor Knockout Mice .” The FASEB Journal 22(8):2676–89. doi: 10.1096/fj.08-105726.
Maiuri, Paolo, Anna Knezevich, Alex De Marco, Davide Mazza, Anna Kula, Jim G. McNally, and Alessandro Marcello. 2011. “Fast Transcription Rates of RNA Polymerase II in Human Cells.” EMBO Reports 12(12):1280–85. doi: 10.1038/embor.2011.196.
Mora, Gloria R., and Virendra B. Mahesh. 1999. Autoregulation of the Androgen Receptor at the Translational Level: Testosterone Induces Accumulation of Androgen Receptor MRNA in the Rat Ventral Prostate Polyribosomes.
Peng, Yajie, Hui Zhu, Bing Han, Yue Xu, Xuemeng Liu, Huaidong Song, and Jie Qiao. 2021. “Identification of Potential Genes in Pathogenesis and Diagnostic Value Analysis of Partial Androgen Insensitivity Syndrome Using Bioinformatics Analysis.” Frontiers in Endocrinology 12. doi: 10.3389/fendo.2021.731107.
Rana, Kesha, Barbara C. Fam, Michele V Clarke, Tammy P. S. Pang, Jeffrey D. Zajac, and Helen E. Maclean. 2011. “Increased Adiposity in DNA Binding-Dependent Androgen Receptor Knockout Male Mice Associated with Decreased Voluntary Activity and Not Insulin Resistance.” Am J Physiol Endocrinol Me-Tab 301:767–78. doi: 10.1152/ajpendo.00584.2010.-In.
Roy, Arun K., Rakesh K. Tyagi, Chung S. Song, Yan Lavrovsky, Soon C. Ahn, Tae Sung Oh, and Bandana Chatterjee. 2001. “Androgen Receptor: Structural Domains and Functional Dynamics after Ligand-Receptor Interaction.” Pp. 44–57 in Annals of the New York Academy of Sciences. Vol. 949. New York Academy of Sciences.
Russell, Patricia K., Michele V. Clarke, Jarrod P. Skinner, Tammy P. S. Pang, Jeffrey D. Zajac, and Rachel A. Davey. 2012. “Identification of Gene Pathways Altered by Deletion of the Androgen Receptor Specifically in Mineralizing Osteoblasts and Osteocytes in Mice.” Journal of Molecular Endocrinology 49(1):1–10. doi: 10.1530/JME-12-0014.
Shiina, Hiroko, Takahiro Matsumoto, Takashi Sato, Katsuhide Igarashi, Junko Miyamoto, Sayuri Takemasa, Matomo Sakari, Ichiro Takada, Takashi Nakamura, Daniel Metzger, Pierre Chambon, Jun Kanno, Hiroyuki Yoshikawa, and Shigeaki Kato. 2006. Premature Ovarian Failure in Androgen Receptor-Deficient Mice. Vol. 103.
Supakar, P. C., C. S. Song, M. H. Jung, M. A. Slomczynska, J. M. Kim, R. L. Vellanoweth, B. Chatterjee, and A. K. Roy. 1993. “A Novel Regulatory Element Associated with Age-Dependent Expression of the Rat Androgen Receptor Gene.” Journal of Biological Chemistry 268(35):26400–408. doi: 10.1016/s0021-9258(19)74328-2.
Tut, Thein G., Farid J. Ghadessy, M. A. Trifiro, L. Pinsky, and E. L. Yong. 1997. Long Polyglutamine Tracts in the Androgen Receptor Are Associated with Reduced Trans-Activation, Impaired Sperm Production, and Male Infertility*. Vol. 82.
Wang, Ruey Sheng, Shuyuan Yeh, Lu Min Chen, Hung Yun Lin, Caixia Zhang, Jing Ni, Cheng Chia Wu, P. Anthony Di Sant’Agnese, Karen L. DeMesy-Bentley, Chii Ruey Tzeng, and Chawnshang Chang. 2006. “Androgen Receptor in Sertoli Cell Is Essential for Germ Cell Nursery and Junctional Complex Formation in Mouse Testes.” Endocrinology 147(12):5624–33. doi: 10.1210/en.2006-0138.
Welsh, M., L. Moffat, K. Belling, L. R. de França, T. M. Segatelli, P. T. K. Saunders, R. M. Sharpe, and L. B. Smith. 2012. “Androgen Receptor Signalling in Peritubular Myoid Cells Is Essential for Normal Differentiation and Function of Adult Leydig Cells.” International Journal of Andrology 35(1):25–40. doi: 10.1111/j.1365-2605.2011.01150.x.
Willems, Ariane, Sergio R. Batlouni, Arantza Esnal, Johannes V. Swinnen, Philippa T. K. Saunders, Richard M. Sharpe, Luiz R. França, Karel de Gendt, and Guido Verhoeven. 2010. “Selective Ablation of the Androgen Receptor in Mouse Sertoli Cells Affects Sertoli Cell Maturation, Barrier Formation and Cytoskeletal Development.” PLoS ONE 5(11). doi: 10.1371/journal.pone.0014168.
Wu, D. I., Grace Lin, and Andrea C. Gore. 2009. “Age-Related Changes in Hypothalamic Androgen Receptor and Estrogen Receptor in Male Rats.” The Journal of Comparative Neurology 512:688–701. doi: 10.1002/cne.21925.
Yu, I. Chen, Hung Yun Lin, Ning Chun Liu, Ruey Shen Wang, Janet D. Sparks, Shuyuan Yeh, and Chawnshang Chang. 2008. “Hyperleptinemia without Obesity in Male Mice Lacking Androgen Receptor in Adipose Tissue.” Endocrinology 149(5):2361–68. doi: 10.1210/en.2007-0516.
Yu, Shengqiang, Chiuan Ren Yeh, Yuanjie Niu, Hong Chiang Chang, Yu Chieh Tsai, Harold L. Moses, Chih Rong Shyr, Chawnshang Chang, and Shuyuan Yeh. 2012. “Altered Prostate Epithelial Development in Mice Lacking the Androgen Receptor in Stromal Fibroblasts.” Prostate 72(4):437–49. doi: 10.1002/pros.21445.
Zhang, Caixia, Shuyuan Yeh, Yen-Ta Chen, Cheng-Chia Wu, Kuang-Hsiang Chuang, Hung-Yun Lin, Ruey-Sheng Wang, Yu-Jia Chang, Chamindrani Mendis-Handagama, Liquan Hu, Henry Lardy, Chawnshang Chang, and † † George. 2006. Oligozoospermia with Normal Fertility in Male Mice Lacking the Androgen Receptor in Testis Peritubular Myoid Cells.
Zhou, Wei, Gensheng Wang, Christopher L. Small, Zhilin Liu, Connie C. Weng, Lizhong Yang, Michael D. Griswold, and Marvin L. Meistrich. 2011. “Erratum: Gene Expression Alterations by Conditional Knockout of Androgen Receptor in Adult Sertoli Cells of Utp14bjsd/Jsd (Jsd) Mice (Biology of Reproduction (2010) 83, (759-766) DOI: 10.1095/Biolreprod.110.085472).” Biology of Reproduction 84(2):400–408.
List of Non Adjacent Key Event Relationships
Relationship: 2828: Decrease, AR activation leads to Hypospadias
AOPs Referencing Relationship
| AOP Name | Adjacency | Weight of Evidence | Quantitative Understanding |
|---|---|---|---|
| Androgen receptor (AR) antagonism leading to hypospadias in male (mammalian) offspring | non-adjacent | High | |
| Decreased testosterone synthesis leading to hypospadias in male (mammalian) offspring | non-adjacent | ||
| 5α-reductase inhibition leading to hypospadias in male (mammalian) offspring | non-adjacent |
Evidence Supporting Applicability of this Relationship
| Life Stage | Evidence |
|---|---|
| Foetal | High |
| Sex | Evidence |
|---|---|
| Male | High |
Taxonomic applicability
In mammals, androgens are one of the primary drivers of penis differentiation. Hypospadias has been observed in several mammals, but most frequently reported in laboratory rodents and in humans (Chang et al., 2020; S. Wang & Zheng, 2025). In vivo studies in rats and mice show that in utero exposure to anti-androgenic chemicals can cause hypospadias in male offspring (see table 3). Many human case studies report boys born with hypospadias and associated deficiency in steroid hormone synthesis, 5α-reductase activity, or androgen receptor (AR) activity (see table 4).
The biologically plausible domain of applicability may extend beyond the empirical domain because androgen-controlled development of male external genitalia is evolutionary conserved in most mammals and, to some extent, also in other vertebrate classes (Gredler et al., 2014). Hypospadias can in principle occur in all animals that form a genital tubercle and have been observed in many domestic animal species including dog (Sonne et al., 2008; Switonski et al., 2018), cat (Nowacka-Woszuk et al., 2014), cattle (Murakami, 2008), sheep (Smith et al., 2012), and horse (De Lorenzi et al., 2010) as well as in wildlife species such as polar bear (Stamper et al., 1999), giraffe (Meuffels et al., 2020), and Tamar Wallaby (Leihy et al., 2011). The observed hypospadias in these animals is not, per se, linked to anti-androgenic exposure, which has only been sparsely investigated in other species than mice, rats, and humans. One study in monkeys did show hypospadias upon oral exposure to finasteride (Prahalada et al., 1997), and bicalutamide exposure induced hypospadias in guinea pigs (S. Wang et al., 2018). A study in rabbits exposed to procymidone did not find hypospadias in males (Inawaka et al., 2010). Another study in hyenas did also not find hypospadias in males after exposure to the anti-androgen finasteride (Drea et al., 1998), but it should be noted that the hyenas have a remarkable sexual development where penile growth occur in both females and males before androgen synthesis is initiated (Cunha et al., 2014) (the studies in hyena and rabbit were identified in our evidence collection but were judged as ‘unreliable’ and therefore not included as empirical evidence).
Sex applicability
The androgen receptor is expressed in the fetal genital tubercle of both females and males (Amato & Yao, 2021; Baskin et al., 2020), but hypospadias is primarily a term used for a malformation of the penis (Baskin & Ebbers, 2006), limiting the applicability of this KER to males.
Life stage applicability
Differentiation of the penis occurs during fetal life in the masculinization programming window (MPW) (GD 16-20 in rats, around gestational weeks 8-14 in humans), when androgen production is high (Welsh et al., 2008; C. Wolf et al., 2000a). In rats, exposure to anti-androgenic chemicals outside of, or in the late part of the MPW does not cause hypospadias or only to a low degree (Clark et al., 1993; van den Driesche et al., 2017; C. Wolf et al., 2000a), while exposure in the earlier (or full) MPW causes a higher frequency of hypospadias (depending on dose and chemical) (table 3). In humans, hypospadias can be diagnosed at birth (X. Yu et al., 2019), while in rodents, some parts of penis development occur postnatally (Schlomer et al., 2013; Sinclair et al., 2017). In these species, hypospadias may be observed at birth but is optimally diagnosed and severity classified weeks later. Given that disruptions to androgen programming takes place in fetal life, even though the AO is best detected postnatally, the life stage applicability is defined as fetal life.
Key Event Relationship Description
This non-adjacent KER describes a fetal decrease in androgen receptor (AR) activation in the genital tubercle causing hypospadias in male offspring, postnatally. During fetal development, androgens induce differentiation of the bipotential genital tubercle to a penis, including closure of the urethra. Androgens signal through AR and reduced fetal AR activation can therefore disrupt penis differentiation and lead to the genital malformation hypospadias. Reduced AR activation may happen both through reduced ligand availability (testosterone or dihydrotestosterone (DHT)) and by direct antagonism of AR (Amato et al., 2022; Mattiske & Pask, 2021).
The upstream KE ‘decrease, androgen receptor activation’ (KE 1614) refers to the in vivo event of overall reduction in AR activation. In this case, it therefore refers to a reduction in AR activation in the genital tubercle. Currently, decreased AR activation in mammals is only directly measured in vitro and not in vivo. Instead, indirect assessment of this KE may come from assays measuring AR antagonism, 5α-reductase activity (the enzyme converting testosterone to DHT), or decreased androgen levels (Draskau et al., 2024).
Evidence Supporting this KER
Biological PlausibilityThe biological plausibility for this KER is judged as high. This is largely based on canonical knowledge on normal reproductive development.
The penis originates from a sexually bipotential structure, the genital tubercle, which may differentiate to either a penis or a clitoris, depending on internal cues during fetal development. In males, the fetal testes produce large amounts of testosterone, which can subsequently be converted to the more potent androgen DHT by 5α-reductase in peripheral tissues. Testosterone and DHT both signal through AR in target tissues to initiate masculinization (Amato et al., 2022; Murashima et al., 2015). The critical developmental window for androgen programming of masculinization has been identified in rats as GD16-20, and is proposed to be gestational weeks 8-14 in humans (Sharpe, 2020; Welsh et al., 2008). As part of the masculinization process orchestrated by androgens, the genital tubercle differentiates to a penis, which at this point expresses AR in both humans and rodents (Amato & Yao, 2021; Baskin et al., 2020). This includes androgen-mediated elongation of the tubercle, formation of the prepuce, and tubular internalization of urethra, which is closed at the distal tip of the glans penis (Amato et al., 2022). Failure of full closure of the urethra can result in hypospadias, in which the urethra terminates at the ventral side of the penis instead of at the tip (Baskin & Ebbers, 2006; Cohn, 2011).
The dependency of androgens for penile development has been demonstrated in mice with conditional or full knockout of Ar, which results in partly or full sex-reversal of males, including a female-like urethral opening (Willingham et al., 2006; Yucel et al., 2004; Zheng et al., 2015). Similarly, female rats and mice exposed in utero to testosterone present with varying degrees of intersexuality, including, in some cases, a penis (Greene & Ivy, 1937; Zheng et al., 2015).
Empirical EvidenceThe empirical evidence for this KER is generally judged high. This includes evidence from in vivo animal studies and evidence from studies in humans. The upstream KE ‘Decreased AR activity’ refers to an in vivo effect, for which no methods for measurement of this in vivo in mammals currently exist. The effects on the upstream KE were therefore indirectly informed as described in each section.
Animal studies
Effects on the upstream KE were indirectly informed by including animal studies with stressors that are known to reduce AR activity through antagonizing the AR, lowering testosterone production, or inhibiting 5α-reductase. Six stressors, with established anti-androgenic effects, were included (more detailed evaluation of these chemicals can be found in KER-2820 (Holmer et al., 2024)). Table 3 summarizes the empirical evidence and confidence level for each chemical. Details on included evidence is presented in Table 1 in Appendix 2, 9prbqyba2x_Appendix_2_KER_2828.pdf. In summary, all six substances were shown to cause hypospadias in male offspring, and the confidence level for all substances was judged as strong, as conflicting results could be explained (see the section ‘Uncertainties and inconsistencies’). Thus, antagonism or AR, inhibition of 5α-reductase, or reduction in testosterone synthesis, all lead to hypospadias.
Table 3 Summary of empirical evidence for the KER – animal studies. See Table 1 in Appendix 2 ( 9prbqyba2x_Appendix_2_KER_2828.pdf) for details.
|
Chemical |
Upstream effect |
Downstream effect |
Overall confidence |
|
Flutamide |
Androgen receptor antagonist (Simard et al., 1986). |
In utero exposure causes hypospadias in rat and mouse |
Strong |
|
Dibutyl phthalate (DBP)
|
Has been shown to reduce fetal intratesticular testosterone and serum testosterone in vivo, but exact mechanism is unknown (Foster, 2006). |
In utero exposure causes hypospadias in rat |
Strong |
|
Vinclozolin |
AR antagonist (Kelce et al., 1994, 1997) |
In utero exposure causes hypospadias in rat and mouse |
Strong |
|
Di(2-ethylhexyl) phthalate (DEHP) |
Has been shown to reduce fetal intratesticular testosterone and serum testosterone in vivo, but exact mechanism is unknown (Parks et al., 2000). |
In utero exposure causes hypospadias in rat |
Strong |
|
Procymidone |
AR antagonist (Ostby et al., 1999). |
In utero exposure causes hypospadias in rat |
Strong |
|
Finasteride |
5α-reductase inhibitor, causing a reduction in DHT (Rittmaster & Wood, 1994). |
In utero exposure causes hypospadias in rat |
Strong |
Supporting human evidence
Effects on the upstream KE were indirectly informed by including studies in humans with a condition (genetic or other) that would reduce or disrupt either 1) function of AR, 2) conversion of testosterone to DHT by disrupting 5α-reductase activity, or 3) production of androgen hormones. Studies measuring low testosterone levels with no underlying cause were also included (see evidence collection strategy). Table 4 lists the studies, in which these conditions were linked to hypospadias in males.
Table 4 Supporting evidence for the KER – human studies. The table lists human studies reporting hypospadias in association with an upstream defect in AR activity, grouped according to the precise effect, and how it was diagnosed (mutation, in vitro activity, or blood hormone and metabolite profile). SRD5A2: 5α-reductase 2; HSD17B3: 17β-hydroxysteroid dehydrogenase 3; HSD3B2: 3β-hydroxysteroid dehydrogenase 2; CYP17A1: 17α-hydroxylase. See table 2 in Appendix 2 (9prbqyba2x_Appendix_2_KER_2828.pdf) for all included references.
|
Effect on upstream KE |
Supporting studies |
|
Effects on Androgen receptor |
|
|
AR mutations |
27 studies |
|
Extended CAG repeat length in AR |
4 studies |
|
Reduced AR activity (e.g. low receptor binding) in in vitro genital skin fibroblasts |
11 studies |
|
Effects on 5α-reductase activity |
|
|
SRD5A2 mutations |
30 studies |
|
SRD5A2 deficiency, diagnosed by T/DHT-ratio and/or reduced in vitro 5α-reductase activity in genital skin fibroblasts |
8 studies |
|
Effects on upstream steroidogenesis enzymes |
|
|
HSD17B3 mutations |
6 studies |
|
HSD3B2 mutations |
5 studies |
|
CYP17A1 mutation |
1 study |
|
HSD17B3 deficiency, diagnosed by hormone and metabolite profile |
2 studies |
|
HSD3B2 deficiency, diagnosed by hormone and metabolite profile |
4 studies |
|
CYP17A1 deficiency, diagnosed by hormone and metabolite profile |
5 studies |
|
Other upstream effects on low testosterone |
|
|
Low testosterone due to gonadal dysgenesis or hypogonadism |
7 studies |
|
Low basal testosterone or low testosterone response to hCG stimulation. Idiopathic or rare mutations. |
7 studies |
Six case-control studies were extracted, all of which found a correlation between lower testosterone levels (basal or hCG-stimulated) and hypospadias (Austin et al., 2002; Okuyama et al., 1981; Raboch et al., 1976; Ratan et al., 2012; Svensson et al., 1979; Yadav et al., 2011). In two of these studies, the correlation was age-dependent (Austin et al., 2002; Raboch et al., 1976)
One epidemiologic study was extracted, which investigated the association between phthalate exposure and hypospadias risk. Western Australian women exposed through their occupation to phthalates were more likely to have sons with hypospadias (Nassar et al., 2010). It should be noted that there are reported species differences in the effects of phthalates (including DEHP and DBP) on fetal testosterone production between humans, mice, and rats, and the direct translatability of the in vivo evidence is uncertain (Sharpe, 2020).
Dose concordance
Direct information about dose concordance is not available because AR activity currently cannot be measured in vivo.
Indirect information on dose concordance can be obtained from empirical evidence. In utero exposure of rats to DBP caused a dose-dependent decrease in serum testosterone levels at PND70 with LOAEL 250 mg/kg bw/day. Hypospadias was observed at this stage with LOAEL 500 mg/kg bw/day (Jiang et al., 2007). It should be noted that fetal testosterone levels were not measured.
Temporal concordance
Direct information about temporal concordance is not available because AR activity currently cannot be measured in vivo.
Indirect information on temporal concordance can be obtained from empirical evidence. In two studies, in which rats were exposed in utero to 750 mg/kg bw/day DBP, intratesticular testosterone levels were reduced in fetal testes, while hypospadias was identified in adult males. Plasma levels of testosterone were also measured in adults, and testosterone levels in exposed males were not significantly different from control males (van den Driesche et al., 2017, 2020). This has also been shown in a study with 500 mg/kg bw/day DBP (Drake et al., 2009). These studies indicate temporal concordance. Another study with DBP-induced hypospadias in rats saw a dose-dependent reduction in serum testosterone levels at PND70 after in utero exposure to as low as 250 mg/kg bw/day from GD14-18 (Jiang et al., 2007), though fetal testosterone levels were not measured in this study.
Incidence Concordance
Direct information about dose concordance is not available because AR activity currently cannot be measured in vivo.
Indirect information on incidence concordance can be obtained from empirical evidence. In the dose-response study with DBP, the incidence of hypospadias was 6.8% for 500 mg/kg bw/day DBP and 41.3% for 750 mg/kg bw/day. When separating hypospadias males from exposed males without hypospadias, plasma testosterone levels were decreased in both groups, indicating that DBP reduced testosterone levels at higher incidence than hypospadias (Jiang et al., 2007). The same was seen in another study with DBP, in which serum testosterone levels at PND7 were reduced in both hypospadiac and non-malformed males exposed to 750 mg/kg bw/day DBP from GD14-18 (Jiang et al., 2016).
Uncertainties and InconsistenciesThe in vivo studies do not directly inform about the upstream KE, ‘decreased AR activity’. The direct concordance between the KEs can therefore not be determined from the evidence.
For flutamide, two studies reported 100% hypospadias frequencies at doses of 6.25 and 10 mg/kg bw/day (Goto et al., 2004; McIntyre et al., 2001), while another study found a frequency of 56.9% when giving 20 mg/kg bw/day (Kita et al., 2016). This might be explained by a longer exposure window in the first two studies and uncertainties in assessment of hypospadias.
For DBP, there were discrepancies in whether 250 mg/kg bw/day was LOAEL (Mylchreest et al., 1998, 1999) or NOAEL (Jiang et al., 2007) for DBP. This conflict was explained by differences in exposure windows, supported by the observation that the frequency of hypospadias at 250 mg/kg bw/day was reported as very low (Mylchreest et al., 1998, 1999).
One study with vinclozolin (Ostby J et al., 1999) and one with procymidone (Hass et al., 2012) did not find hypospadias after in utero exposure. In both cases, this was likely due to too low doses tested.
In most of the human studies of steroidogenesis deficiency, serum or plasma levels of testosterone were reduced at baseline and/or upon hCG stimulation (Al-Sinani et al., 2015; Ammini et al., 1997; Cara et al., 1985; Chen, Huang, et al., 2021; Dean et al., 1984; Galli-Tsinopoulou et al., 2018; Imperato-McGinley et al., 1979; Kaufman et al., 1983; Mendonca et al., 1987, 2000; Neocleous et al., 2012; New, 1970; Pang et al., 1983; Perrone et al., 1985; Rabbani et al., 2012; Sherbet et al., 2003), but in a few studies, testosterone levels were normal (Donadille et al., 2018; Kon et al., 2015; Luna et al., 2021). In these cases, the effect of these deficiencies on tissue AR activity is uncertain.
For AR CAG repeat length, a case-control study did not find an association with hypospadias (Radpour R et al., 2007), but this could be because the hypospadias cases included had other etiologies.
Lastly, as there are currently no universal guidelines for identification and scoring of hypospadias in rodents, there are large variations in methods of assessment, and minor cases of hypospadias may be overlooked in some studies and included in others. This poses an uncertainty in the frequency reports in the scientific evidence.
Quantitative Understanding of the Linkage
The quantitative understanding of the relationship is low. As there are currently no direct measurement methods of the upstream KE (reduced AR activity) in mammals, quantification of the relationship is difficult to assess.
Response-response relationshipA model for phthalates has been developed, aiming to predict the frequency of hypospadias in male offspring based on reductions in ex vivo testosterone production, an indirect indication of AR activity. In this model, hypospadias was induced from around a 60% reduction in testosterone levels. The model does not consider hypospadias severity and is only for phthalate chemicals (Earl Gray et al., 2024).
Time-scaleThe time-scale of this KER depends on the species but is likely days to weeks.
AR activation happens within minutes, from ligand binding to nuclear translocation and promotor activation (Nightingale et al., 2003; Schaufele et al., 2005), while transcriptional and translational effects are observed minutes to hours later (Kang et al., 2002). AR programming of the genital tubercle occurs during fetal development in the Masculinization Programming Window (Sharpe, 2020). The time-scale for morphological effects in the tissue then depends on the species. In humans, penis development is completed prior to birth and hypospadias can be observed at birth. In rodents, penis development is not fully completed until weeks after birth, but hypospadias can often be observed earlier than this (table 3).
Known modulating factors|
Modulating Factors |
MF details |
Effects on the KER |
References |
|
AR CAG repeat length |
Extended CAG repeat length in AR is associated with reduced AR activity |
Higher risk of hypospadias development |
(Chamberlain et al., 1994) |
There are no known feedforward/feedback loops influencing this KER.
References
Al-Sinani, A., Mula-Abed, W., Al-Kindi, M., Al-Kusaibi, G., Al-Azkawi, H., & Nahavandi, N. (2015). A Novel Mutation Causing 17-β-Hydroxysteroid Dehydrogenase Type 3 Deficiency in an Omani Child: First Case Report and Review of Literature. Oman Medical Journal, 30(2), 129–134. https://doi.org/10.5001/omj.2015.27
Amato, C. M., & Yao, H. H.-C. (2021). Developmental and sexual dimorphic atlas of the prenatal mouse external genitalia at the single-cell level. Proceedings of the National Academy of Sciences of the United States of America, 118(25). https://doi.org/10.1073/pnas.2103856118
Amato, C. M., Yao, H. H.-C., & Zhao, F. (2022). One Tool for Many Jobs: Divergent and Conserved Actions of Androgen Signaling in Male Internal Reproductive Tract and External Genitalia. Frontiers in Endocrinology, 13, 910964. https://doi.org/10.3389/fendo.2022.910964
Ammini, A., Sharma, D., Gupta, R., Mohapatra, I., Kucheria, K., Kriplani, A., Takkar, D., Mitra, D., & Vijayaraghavan, M. (1997). Familial male pseudohermaphroditism. Indian Journal of Pediatrics, 64(3), 419–423. https://doi.org/10.1007/BF02845218
Austin, P., Siow, Y., Fallat, M., Cain, M., Rink, R., & Casale, A. (2002). The relationship between müllerian inhibiting substance and androgens in boys with hypospadias. The Journal of Urology, 168(4), 1784–1788; discussion 1788. https://doi.org/10.1097/01.ju.0000023680.64155.5c
Baskin, L., Cao, M., Sinclair, A., Li, Y., Overland, M., Isaacson, D., & Cunha, G. R. (2020). Androgen and estrogen receptor expression in the developing human penis and clitoris. Differentiation; Research in Biological Diversity, 111, 41–59. https://doi.org/10.1016/j.diff.2019.08.005
Baskin, L., & Ebbers, M. (2006). Hypospadias: Anatomy, etiology, and technique. Journal of Pediatric Surgery, 41(3), 463–472. https://doi.org/10.1016/j.jpedsurg.2005.11.059
Cara, J., Jr Moshang, T., Bongiovanni, A., & Marx, B. (1985). Elevated 17-hydroxyprogesterone and testosterone in a newborn with 3-beta-hydroxysteroid dehydrogenase deficiency. The New England Journal of Medicine, 313(10), 618–621. https://doi.org/10.1056/NEJM198509053131007
Chamberlain, N. L., Driver, E. D., & Miesfeld, R. L. (1994). The length and location of CAG trinucleotide repeats in the androgen receptor N-terminal domain affect transactivation function. Nucleic Acids Research, 22(15), 3181–3186. https://doi.org/10.1093/nar/22.15.3181
Chang, J., Wang, S., & Zheng, Z. (2020). Etiology of Hypospadias: A Comparative Review of Genetic Factors and Developmental Processes Between Human and Animal Models. Research and Reports in Urology, Volume 12, 673–686. https://doi.org/10.2147/RRU.S276141
Chen, L., Huang, H., Zhang, H., Zhu, G., & Zhu, M. (2021). Three cases of 3β-hydroxysteroid dehydrogenase deficiency: Clinical analysis. Advances in Clinical and Experimental Medicine : Official Organ Wroclaw Medical University, 30(3), 289–299. https://doi.org/10.17219/acem/131220
Clark, R., Anderson, C., Prahalada, S., Robertson, R., Lochry, E., Leonard, Y., Stevens, J., & Hoberman, A. (1993). Critical developmental periods for effects on male rat genitalia induced by finasteride, a 5 alpha-reductase inhibitor. Toxicology and Applied Pharmacology, 119(1), 34–40. https://doi.org/10.1006/taap.1993.1041
Cohn, M. J. (2011). Development of the external genitalia: Conserved and divergent mechanisms of appendage patterning. Developmental Dynamics, 240(5), 1108–1115. https://doi.org/10.1002/dvdy.22631
Cunha, G. R., Risbridger, G., Wang, H., Place, N. J., Grumbach, M., Cunha, T. J., Weldele, M., Conley, A. J., Barcellos, D., Agarwal, S., Bhargava, A., Drea, C., Hammond, G. L., Siiteri, P., Coscia, E. M., McPhaul, M. J., Baskin, L. S., & Glickman, S. E. (2014). Development of the external genitalia: Perspectives from the spotted hyena (Crocuta crocuta). Differentiation, 87(1–2), 4–22. https://doi.org/10.1016/j.diff.2013.12.003
De Lorenzi, L., Genualdo, V., Iannuzzi, A., Di Meo, G. P., Perucatti, A., Mancuso, R., Russo, M., Di Berardino, D., Parma, P., & Iannuzzi, L. (2010). Cytogenetic and Genetic Studies in a Hypospadic Horse (Equus caballus, 2n = 64). Sexual Development, 4(6), 352–357. https://doi.org/10.1159/000319527
Dean, H., Shackleton, C., & Winter, J. (1984). Diagnosis and natural history of 17-hydroxylase deficiency in a newborn male. The Journal of Clinical Endocrinology and Metabolism, 59(3), 513–520. https://doi.org/10.1210/jcem-59-3-513
Donadille, B., Houang, M., Netchine, I., Siffroi, J., & Christin-Maitre, S. (2018). Human 3beta-hydroxysteroid dehydrogenase deficiency associated with normal spermatic numeration despite a severe enzyme deficit. Endocrine Connections, 7(3), 395–402. https://doi.org/10.1530/EC-17-0306
Drake, A., van den Driesche, S., Scott, H., Hutchison, G., Seckl, J., & Sharpe, R. (2009). Glucocorticoids amplify dibutyl phthalate-induced disruption of testosterone production and male reproductive development. Endocrinology, 150(11), 5055–5064. https://doi.org/10.1210/en.2009-0700
Draskau, M. K., Rosenmai, A. K., Bouftas, N., Johansson, H. K. L., Panagiotou, E. M., Holmer, M. L., Elmelund, E., Zilliacus, J., Beronius, A., Damdimopolou, P., van Duursen, M., & Svingen, T. (2024). AOP Report: An Upstream Network for Reduced Androgen Signaling Leading to Altered Gene Expression of Androgen Receptor-Responsive Genes in Target Tissues. Environmental Toxicology and Chemistry. https://doi.org/10.1002/etc.5972
Drea, C., Weldele, M., Forger, N., Coscia, E., Frank, L., Licht, P., & Glickman, S. (1998). Androgens and masculinization of genitalia in the spotted hyaena (Crocuta crocuta). 2. Effects of prenatal anti-androgens. Journal of Reproduction and Fertility, 113(1), 117–127. https://doi.org/10.1530/jrf.0.1130117
Earl Gray, L. J., Lambright, C., Evans, N., Ford, J., & Conley, M. (2024). Using targeted fetal rat testis genomic and endocrine alterations to predict the effects of a phthalate mixture on the male reproductive tract. Current Research in Toxicology, 7, 100180. https://doi.org/10.1016/j.crtox.2024.100180
Foster, P. M. D. (2006). Disruption of reproductive development in male rat offspring following in utero exposure to phthalate esters. International Journal of Andrology, 29(1), 140–147. https://doi.org/10.1111/j.1365-2605.2005.00563.x
Galli-Tsinopoulou, A., Serbis, A., KotanidouP, E., Litou, E., Dokousli, V., Mouzaki, K., Fanis, P., Neocleous, V., & Skordis, N. (2018). 46,XY Disorder of Sex Development due to 17-Beta Hydroxysteroid Dehydrogenase Type 3 Deficiency in an Infant of Greek Origin. Journal of Clinical Research in Pediatric Endocrinology, 10(1), 74–78. https://doi.org/10.4274/jcrpe.4829
Goto, K., Koizumi, K., Takaori, H., Fujii, Y., Furuyama, Y., Saika, O., Suzuki, H., Saito, K., & Suzuki, K. (2004). Effects of flutamide on sex maturation and behavior of offspring born to female rats treated during late pregnancy. The Journal of Toxicological Sciences, 29(5), 517–534. https://doi.org/10.2131/jts.29.517
Gredler, M. L., Larkins, C. E., Leal, F., Lewis, A. K., Herrera, A. M., Perriton, C. L., Sanger, T. J., & Cohn, M. J. (2014). Evolution of External Genitalia: Insights from Reptilian Development. Sexual Development, 8(5), 311–326. https://doi.org/10.1159/000365771
Greene, R. R., & Ivy, A. C. (1937). THE EXPERIMENTAL PRODUCTION OF INTERSEXUALITY IN THE FEMALE RAT WITH TESTOSTERONE. Science (New York, N.Y.), 86(2226), 200–201. https://doi.org/10.1126/science.86.2226.200-a
Hass, U., Boberg, J., Christiansen, S., Jacobsen, P., Vinggaard, A., Taxvig, C., Poulsen, M., Herrmann, S., Jensen, B., Petersen, A., Clemmensen, L., & Axelstad, M. (2012). Adverse effects on sexual development in rat offspring after low dose exposure to a mixture of endocrine disrupting pesticides. Reproductive Toxicology (Elmsford, N.Y.), 34(2), 261–274. https://doi.org/10.1016/j.reprotox.2012.05.090
Holmer, M. L., Zilliacus, J., Draskau, M. K., Hlisníková, H., Beronius, A., & Svingen, T. (2024). Methodology for developing data-rich Key Event Relationships for Adverse Outcome Pathways exemplified by linking decreased androgen receptor activity with decreased anogenital distance. Reproductive Toxicology, 128, 108662. https://doi.org/10.1016/j.reprotox.2024.108662
Imperato-McGinley, J., Peterson, R., Stoller, R., & Goodwin, W. (1979). Male pseudohermaphroditism secondary to 17 beta-hydroxysteroid dehydrogenase deficiency: Gender role change with puberty. The Journal of Clinical Endocrinology and Metabolism, 49(3), 391–395. https://doi.org/10.1210/jcem-49-3-391
Inawaka, K., Kishimoto, N., Higuchi, H., & Kawamura, S. (2010). Maternal exposure to procymidone has no effects on fetal external genitalia development in male rabbit fetuses in a modified developmental toxicity study. JOURNAL OF TOXICOLOGICAL SCIENCES, 35(3), 299–307. https://doi.org/10.2131/jts.35.299
Jiang, J., Ma, L., Yuan, L., Wang, X., & Zhang, W. (2007). Study on developmental abnormalities in hypospadiac male rats induced by maternal exposure to di-n-butyl phthalate (DBP). Toxicology, 232(3), 286–293. https://doi.org/10.1016/j.tox.2007.01.018
Jiang, J., Zhong, C., Zhu, Y., XuL, D., Wood, K., Sun, W., Li, E., Liu, Z., Zhao, W., Ruan, Y., & Xia, S. (2016). Prenatal exposure to di-n-butyl phthalate (DBP) differentially alters androgen cascade in undeformed versus hypospadiac male rat offspring. Reproductive Toxicology (Elmsford, N.Y.), 61, 75–81. https://doi.org/10.1016/j.reprotox.2016.02.016
Kang, H.-Y., Huang, K.-E., Chang, S. Y., Ma, W.-L., Lin, W.-J., & Chang, C. (2002). Differential Modulation of Androgen Receptor-mediated Transactivation by Smad3 and Tumor Suppressor Smad4. Journal of Biological Chemistry, 277(46), 43749–43756. https://doi.org/10.1074/jbc.M205603200
Kaufman, F., Costin, G., Goebelsmann, U., Stanczyk, F., & Zachmann, M. (1983). Male pseudohermaphroditism due to 17,20-desmolase deficiency. The Journal of Clinical Endocrinology and Metabolism, 57(1), 32–36. https://doi.org/10.1210/jcem-57-1-32
Kelce, W. R., Lambright, C. R., Gray, L. E., & Roberts, K. P. (1997). Vinclozolin andp,p′-DDE Alter Androgen-Dependent Gene Expression:In VivoConfirmation of an Androgen Receptor-Mediated Mechanism. Toxicology and Applied Pharmacology, 142(1), 192–200. https://doi.org/10.1006/taap.1996.7966
Kelce, W. R., Monosson, E., Gamcsik, M. P., Laws, S. C., & Gray, L. E. (1994). Environmental Hormone Disruptors: Evidence That Vinclozolin Developmental Toxicity Is Mediated by Antiandrogenic Metabolites. Toxicology and Applied Pharmacology, 126(2), 276–285. https://doi.org/10.1006/taap.1994.1117
Kita, D., Meyer, K., Venturelli, A., Adams, R., Machado, D., Morais, R., Swan, S., Gennings, C., & Martino-Andrade, A. (2016). Manipulation of pre and postnatal androgen environments and anogenital distance in rats. TOXICOLOGY, 368, 152–161. https://doi.org/10.1016/j.tox.2016.08.021
Kon, M., Suzuki, E., Dung, V., Hasegawa, Y., Mitsui, T., Muroyas, K., Ueoka, K., Igarashi, N., Nagasaki, K., Oto, Y., Hamajima, T., Yoshino, K., Igarashi, M., Kato-Fukui, Y., Nakabayashi, K., Hayashi, K., Hata, K., Matsubara, Y., Moriya, K., … Fukami, M. (2015). Molecular basis of non-syndromic hypospadias: Systematic mutation screening and genome-wide copy-number analysis of 62 patients. HUMAN REPRODUCTION, 30(3), 499–506. https://doi.org/10.1093/humrep/deu364
Leihy, M. W., Shaw, G., Wilson, J. D., & Renfree, M. B. (2011). Development of the Penile Urethra in the Tammar Wallaby. Sexual Development, 5(5), 241–249. https://doi.org/10.1159/000334053
Luna, S., Wegner, D., Gale, S., Yang, P., Hollander, A., St Dennis-Feezle, L., Nabhan, Z., Ory, D., Cole, F., & Wambach, J. (2021). Whole exome sequencing and functional characterization increase diagnostic yield in siblings with a 46, XY difference of sexual development (DSD). The Journal of Steroid Biochemistry and Molecular Biology, 212, 105908. https://doi.org/10.1016/j.jsbmb.2021.105908
Mattiske, D. M., & Pask, A. J. (2021). Endocrine disrupting chemicals in the pathogenesis of hypospadias; developmental and toxicological perspectives. Current Research in Toxicology, 2, 179–191. https://doi.org/10.1016/j.crtox.2021.03.004
McIntyre, B., Barlow, N., & Foster, P. (2001). Androgen-mediated development in male rat offspring exposed to flutamide in utero: Permanence and correlation of early postnatal changes in anogenital distance and nipple retention with malformations in androgen-dependent tissues. Toxicological Sciences : An Official Journal of the Society of Toxicology, 62(2), 236–249. https://doi.org/10.1093/toxsci/62.2.236
Mendonca, B., Bloise, W., Arnhold, I., Batista, M., Toledo, S., Drummond, M., Nicolau, W., & Mattar, E. (1987). Male pseudohermaphroditism due to nonsalt-losing 3 beta-hydroxysteroid dehydrogenase deficiency: Gender role change and absence of gynecomastia at puberty. Journal of Steroid Biochemistry, 28(6), 669–675. https://doi.org/10.1016/0022-4731(87)90396-7
Mendonca, B., Inacio, M., Arnhold, I., Costa, E., Bloise, W., Martin, R., Denes, F., Silva, F., Andersson, S., Lindqvist, A., & Wilson, J. (2000). Male pseudohermaphroditism due to 17 beta-hydroxysteroid dehydrogenase 3 deficiency. Diagnosis, psychological evaluation, and management. Medicine, 79(5), 299–309. https://doi.org/10.1097/00005792-200009000-00003
Meuffels, J., Luther-Binoir, I., Daffue, W., Deacon, F., & Mitchell, E. P. (2020). Testicular disorder of sexual development with cryptorchidism, penile hypoplasia and hypospadias in a giraffe (Giraffa camelopardalis giraffa). JOURNAL OF THE SOUTH AFRICAN VETERINARY ASSOCIATION, 91. https://doi.org/10.4102/jsava.v91i0.1971
Murakami, T. (2008). Anatomical Examination of Hypospadias in Cattle. Journal of the Japan Veterinary Medical Association, 61(12), 931–935. https://doi.org/10.12935/jvma1951.61.931
Murashima, A., Kishigami, S., Thomson, A., & Yamada, G. (2015). Androgens and mammalian male reproductive tract development. Biochimica et Biophysica Acta (BBA) - Gene Regulatory Mechanisms, 1849(2), 163–170. https://doi.org/10.1016/j.bbagrm.2014.05.020
Mylchreest, E., Cattley, R., & Foster, P. (1998). Male reproductive tract malformations in rats following gestational and lactational exposure to Di(n-butyl) phthalate: An antiandrogenic mechanism? Toxicological Sciences : An Official Journal of the Society of Toxicology, 43(1), 47–60. https://doi.org/10.1006/toxs.1998.2436
Mylchreest, E., Sar, M., Cattley, R., & Foster, P. (1999). Disruption of androgen-regulated male reproductive development by di(n-butyl) phthalate during late gestation in rats is different from flutamide. Toxicology and Applied Pharmacology, 156(2), 81–95. https://doi.org/10.1006/taap.1999.8643
Nassar, N., Abeywardana, P., Barker, A., & Bower, C. (2010). Parental occupational exposure to potential endocrine disrupting chemicals and risk of hypospadias in infants. Occupational and Environmental Medicine, 67(9), 585–589. https://doi.org/10.1136/oem.2009.048272
Neocleous, V., Sismani, C., Shammas, C., Efstathiou, E., Alexandrou, A., Ioannides, M., Argyrou, M., Patsalis, P., Phylactou, L., & Skordis, N. (2012). Duplication of exons 3-10 of the HSD17B3 gene: A novel type of genetic defect underlying 17 beta-HSD-3 deficiency. GENE, 499(2), 250–255. https://doi.org/10.1016/j.gene.2012.03.031
New, M. (1970). Male pseudohermaphroditism due to 17 alpha-hydroxylase deficiency. The Journal of Clinical Investigation, 49(10), 1930–1941. https://doi.org/10.1172/JCI106412
Nightingale, J., Chaudhary, K. S., Abel, P. D., Stubbs, A. P., Romanska, H. M., Mitchell, S. E., Stamp, G. W. H., & Lalani, E.-N. (2003). Ligand Activation of the Androgen Receptor Downregulates E-Cadherin-Mediated Cell Adhesion and Promotes Apoptosis of Prostatic Cancer Cells. Neoplasia, 5(4), 347–361. https://doi.org/10.1016/S1476-5586(03)80028-3
Nowacka-Woszuk, J., Szczerbal, I., Salamon, S., Kociucka, B., Jackowiak, H., Prozorowska, E., Slaska, B., Rozanska, D., Orzelski, M., Ochota, M., Dzimira, S., Lipiec, M., Nizanski, W., & Switonski, M. (2014). Testicular disorder of sex development in four cats with a male karyotype (38,XY; SRY-positive). Animal Reproduction Science, 151(1–2), 42–48. https://doi.org/10.1016/j.anireprosci.2014.10.001
Okuyama, A., Namiki, M., Koide, T., Itatani, H., Nishimoto, N., Mizutani, S., Sonoda, T., Aono, T., & Matsumoto, K. (1981). Pituitary and gonadal function in prepubertal and pubertal boys with hypospadias. Acta Endocrinologica, 98(3), 464–469. https://doi.org/10.1530/acta.0.0980464
Ostby, J., Kelce, W. R., Lambright, C., Wolf, C. J., Mann, P., & Gray, L. E. (1999). The fungicide procymidone alters sexual differentiation in the male rat by acting as an androgen-receptor antagonist in vivo and in vitro. Toxicology and Industrial Health, 15(1–2), 80–93. https://doi.org/10.1177/074823379901500108
Ostby J, Monosson E, Kelce WR, & Gray, L. J. (1999). Environmental antiandrogens: Low doses of the fungicide vinclozolin alter sexual differentiation of the male rat. Toxicology and Industrial Health, 15(1), 48–64. https://doi.org/10.1177/074823379901500106
Pang, S., Levine, L., Stoner, E., Opitz, J., Pollack, M., Dupont, B., & New, M. (1983). Nonsalt-losing congenital adrenal hyperplasia due to 3 beta-hydroxysteroid dehydrogenase deficiency with normal glomerulosa function. The Journal of Clinical Endocrinology and Metabolism, 56(4), 808–818. https://doi.org/10.1210/jcem-56-4-808
Parks, L. G., Ostby, J. S., Lambright, C. R., Abbott, B.D., Klinefelter, G. R., Barlow, N. J., & Gray LE Jr. (2000). The Plasticizer Diethylhexyl Phthalate Induces Malformations by Decreasing Fetal Testosterone Synthesis during Sexual Differentiation in the Male Rat. Toxicological Sciences, 58(2), 339–349. https://doi.org/10.1093/toxsci/58.2.339
Perrone, L., Criscuolo, T., Sinisi, A., Graziani, M., Manzo, T., Sicuranza, R., Bellastella, A., & Faggiano, M. (1985). Male pseudohermaphroditism due to 3 beta-hydroxysteroid dehydrogenase-isomerase deficiency associated with atrial septal defect. Acta Endocrinologica, 110(4), 532–539. https://doi.org/10.1530/acta.0.1100532
Prahalada, S., Tarantal, A., Harris, G., Ellsworth, K., Clarke, A., Skiles, G., MacKenzie, K., Kruk, L., Ablin, D., Cukierski, M., Peter, C., vanZwieten, M., & Hendrickx, A. (1997). Effects of finasteride, a type 2 5-alpha reductase inhibitor, on fetal development in the rhesus monkey (Macaca mulatta). Teratology, 55(2), 119–131. https://doi.org/10.1002/(SICI)1096-9926(199702)55:2%253C119::AID-TERA1%253E3.0.CO;2-Z
Rabbani, B., Mahdieh, N., Ashtiani, M. H., Setoodeh, A., & A Rabbani. (2012). In silico structural, functional and pathogenicity evaluation of a novel mutation: An overview of HSD3B2 gene mutations. Gene, 503(2), 215–221. https://doi.org/10.1016/j.gene.2012.04.080
Raboch, J., Pondelickova, J., & Starka, L. (1976). Plasma testosterone values in hypopspadiacs. Andrologia, 8(3), 255–258. https://doi.org/10.1111/j.1439-0272.1976.tb02144.x
Radpour R, Rezaee M, Tavasoly A, Solati S, & Saleki A. (2007). Association of long polyglycine tracts (GGN repeats) in exon 1 of the androgen receptor gene with cryptorchidism and penile hypospadias in Iranian patients. Journal of Andrology, 28(1), 164–169. https://doi.org/10.2164/jandrol.106.000927
Ratan, S., Aggarwal, S., Mishra, T., Saxena, A., Yadav, S., Pandey, R., Sharma, A., & Dhanwal, D. (2012). Children with isolated hypospadias have different hormonal profile compared to those with associated anomalies. Journal of Pediatric Endocrinology & Metabolism : JPEM, 25(1), 111–119. https://doi.org/10.1515/jpem.2011.421
Rittmaster, R. S., & Wood, A. J. J. (1994). Finasteride. New England Journal of Medicine, 330(2), 120–125. https://doi.org/10.1056/NEJM199401133300208
Schaufele, F., Carbonell, X., Guerbadot, M., Borngraeber, S., Chapman, M. S., Ma, A. A. K., Miner, J. N., & Diamond, M. I. (2005). The structural basis of androgen receptor activation: Intramolecular and intermolecular amino–carboxy interactions. Proceedings of the National Academy of Sciences, 102(28), 9802–9807. https://doi.org/10.1073/pnas.0408819102
Schlomer, B. J., Feretti, M., Rodriguez, E. J., Blaschko, S., Cunha, G., & Baskin, L. (2013). Sexual differentiation in the male and female mouse from days 0 to 21: A detailed and novel morphometric description. The Journal of Urology, 190(4 Suppl), 1610–1617. https://doi.org/10.1016/j.juro.2013.02.3198
Sharpe, R. M. (2020). Androgens and the masculinization programming window: Human-rodent differences. Biochemical Society Transactions, 48(4), 1725–1735. https://doi.org/10.1042/BST20200200
Sherbet, D., Tiosano, D., Kwist, K., Hochberg, Z., & RJ Auchus. (2003). CYP17 mutation E305G causes isolated 17,20-lyase deficiency by selectively altering substrate binding. The Journal of Biological Chemistry, 278(49), 48563–48569. https://doi.org/10.1074/jbc.M307586200
Simard, J., Luthy, I., Guay, J., Bélanger, A., & Labrie, F. (1986). Characteristics of interaction of the antiandrogen flutamide with the androgen receptor in various target tissues. Molecular and Cellular Endocrinology, 44(3), 261–270. https://doi.org/10.1016/0303-7207(86)90132-2
Sinclair, A., Cao, M., Pask, A., Baskin, L., & Cunha, G. (2017). Flutamide-induced hypospadias in rats: A critical assessment. Differentiation; Research in Biological Diversity, 94, 37–57. https://doi.org/10.1016/j.diff.2016.12.001
Smith, K., Brown, P., & Barr, F. (2012). A Survey of Congenital Reproductive Abnormalities in Rams in Abattoirs in South West England. Reproduction in Domestic Animals, 47(5), 740–745. https://doi.org/10.1111/j.1439-0531.2011.01952.x
Sonne, C., Dietz, R., Born, E. W., Leifsson, P. S., & Andersen, S. (2008). Is there a link between hypospadias and organochlorine exposure in East Greenland sledge dogs (Canis familiaris)? Ecotoxicology and Environmental Safety, 69(3), 391–395. https://doi.org/10.1016/j.ecoenv.2007.09.008
Stamper, M. A., Norton, T., Spodnick, G., Marti, J., & Loomis, M. (1999). Hypospadias in a polar bear (Ursus maritimus). Journal of Zoo and Wildlife Medicine: Official Publication of the American Association of Zoo Veterinarians, 30(1), 141–144.
Svensson, J., Eneroth, P., Gustafsson, J., Ritzén, M., & Stenberg, A. (1979). Reduction of androstenedione by skin in vitro and serum levels of gonadotrophins and androgens in men with hypospadias. The Journal of Endocrinology, 82(3), 395–401. https://doi.org/10.1677/joe.0.0820395
Switonski, M., Dzimira, S., Aleksiewicz, R., Szczerbal, I., Nowacka-Woszuk, J., Krzeminska, P., Deska, T., & Nizanski, W. (2018). Hypospadias Is Not Rare in Dogs: Five New Cases, a Retrospective Study, and a Review of the Literature. Sexual Development : Genetics, Molecular Biology, Evolution, Endocrinology, Embryology, and Pathology of Sex Determination and Differentiation, 12(5), 244–250. https://doi.org/10.1159/000490079
van den Driesche, S., Kilcoyne, K., Wagner, I., Rebourcet, D., Boyle, A., Mitchell, R., McKinnell, C., Macpherson, S., Donat, R., Shukla, C., Jorgensen, A., Meyts, E., Skakkebaek, N., & Sharpe, R. (2017). Experimentally induced testicular dysgenesis syndrome originates in the masculinization programming window. JCI Insight, 2(6), e91204. https://doi.org/10.1172/jci.insight.91204
van den Driesche, S., Shoker, S., Inglis, F., Palermo, C., Langsch, A., & Otter, R. (2020). Systematic comparison of the male reproductive tract in fetal and adult Wistar rats exposed to DBP and DINP in utero during the masculinisation programming window. Toxicology Letters, 335, 37–50. https://doi.org/10.1016/j.toxlet.2020.10.006
Wang, S., Shi, M., Zhu, D., Mathews, R., & Zheng, Z. (2018). External Genital Development, Urethra Formation, and Hypospadias Induction in Guinea Pig: A Double Zipper Model for Human Urethral Development. Urology, 113, 179–186. https://doi.org/10.1016/j.urology.2017.11.002
Wang, S., & Zheng, Z. (2025). Differences in Formation of Prepuce and Urethral Groove During Penile Development Between Guinea Pigs and Mice Are Controlled by Differential Expression of Shh, Fgf10 and Fgfr2. Cells, 14(5), 348. https://doi.org/10.3390/cells14050348
Welsh, M., Saunders, P., Fisken, M., Scott, H., Hutchison, G., Smith, L., & Sharpe, R. (2008). Identification in rats of a programming window for reproductive tract masculinization, disruption of which leads to hypospadias and cryptorchidism. The Journal of Clinical Investigation, 118(4), 1479–1490. https://doi.org/10.1172/JCI34241
Willingham, E., Agras, K., Souza, A. J. de, Konijeti, R., Yucel, S., Rickie, W., Cunha, G., & Baskin, L. (2006). Steroid receptors and mammalian penile development: An unexpected role for progesterone receptor? The Journal of Urology, 176(2), 728–733. https://doi.org/10.1016/j.juro.2006.03.078
Wolf, C., LeBlanc, G., Ostby, J., & Gray, L. J. (2000). Characterization of the period of sensitivity of fetal male sexual development to vinclozolin. Toxicological Sciences : An Official Journal of the Society of Toxicology, 55(1), 152–161. https://doi.org/10.1093/toxsci/55.1.152
Yadav, C., Bajpai, M., Kumar, V., Datta, S., Gupta, P., Ahmed, R., & Banerjee, B. (2011). Polymorphisms in the P450 c17 (17-hydroxylase/17, 20-Lyase) gene: Association with estradiol and testosterone concentration in hypospadias. Urology, 78(4), 902–907. https://doi.org/10.1016/j.urology.2011.04.021
Yu, X., Nassar, N., Mastroiacovo, P., Canfield, M., Groisman, B., Bermejo-Sánchez, E., Ritvanen, A., Kiuru-Kuhlefelt, S., Benavides, A., Sipek, A., Pierini, A., Bianchi, F., Källén, K., Gatt, M., Morgan, M., Tucker, D., Canessa, M. A., Gajardo, R., Mutchinick, O. M., … Agopian, A. J. (2019). Hypospadias Prevalence and Trends in International Birth Defect Surveillance Systems, 1980-2010. European Urology, 76(4), 482–490. https://doi.org/10.1016/j.eururo.2019.06.027
Yucel, S., Liu, W., Cordero, D., Donjacour, A., Cunha, G., & Baskin, L. (2004). Anatomical studies of the fibroblast growth factor-10 mutant, Sonic Hedge Hog mutant and androgen receptor mutant mouse genital tubercle. Advances in Experimental Medicine and Biology, 545, 123–148. https://doi.org/10.1007/978-1-4419-8995-6_8
Zheng, Z., Armfield, B., & Cohn, M. (2015). Timing of androgen receptor disruption and estrogen exposure underlies a spectrum of congenital penile anomalies. Proceedings of the National Academy of Sciences of the United States of America, 112(52), E7194-203. https://doi.org/10.1073/pnas.1515981112